-
中药青蒿为植物菊科黄花蒿(Artemisia annua L.)的干燥地上部分,味苦、辛,性寒,归肝、胆经,具有清虚热等功效[1]。现代研究表明,青蒿及其所含有关单体成分除了在抗疟疾方面有效外,在抑菌抗炎、免疫调节和缓解热应激等方面也有效果[2],特别是在抗氧化方面,青蒿亦表现出良好作用[3]。现有研究表明,氧化应激与许多疾病关系密切,过度氧化应激可导致骨质疏松症等疾病[4]。目前,对青蒿所含活性成分青蒿素研究较多,特别是其抗疟、抗氧化等作用受到关注[5],但青蒿素之外的其他成分在抗氧化方面亦有活性[6-9],应受到重视。本研究进行青蒿抗氧化活性的谱效关系研究,分析色谱特征峰与DPPH和ABTS自由基清除能力的关联性[10],探究青蒿所含不同化学成分的抗氧化活性,以阐明青蒿体外抗氧化活性的药效物质基础,为青蒿的质量评价和资源开发等提供依据。
-
岛津LC-20AD型高效液相色谱仪,包括四元泵、PAD检测器、自动进样器(日本岛津株式会社);BT285S型电子分析天平(德国Sartorius 公司);DL-1000B型超声波清洗仪(上海之信仪器有限公司);ELx 800型多功能酶标仪(美国 Biotek公司)。
-
东莨菪内酯(批号:20220215)、芹菜素(批号:20220216)、木犀草素(批号:20220216)购自上海历鼎生物技术有限公司;异绿原酸A(批号:DSTDY054701)、异绿原酸B(批号:DST210625-037)、异绿原酸C(批号:DSTDY003802)、滨蒿内酯(批号:DSTDB003101)、艾黄素(批号:DST220119-004)、青蒿素(批号:DSTDQ004702)、青蒿酸(批号:DST210919-048)购自成都德思特生物技术有限公司;所有对照品纯度均≥98%。DPPH自由基清除能力试剂盒(批号:20220223)、ABTS自由基清除能力试剂盒(批号:20220224)购自上海励瑞生物科技有限公司。
-
11批青蒿药材样品信息见表1,经海军军医大学药学系生药教研室辛海量教授鉴定为Artemisia annua L.的干燥地上部分。
序号 产地 序号 产地 S1 河北邯郸 S7 安徽亳州 S2 安徽亳州 S8 江西赣州 S3 河南驻马店 S9 四川自贡 S4 安徽亳州 S10 河南南阳 S5 湖北恩施 S11 重庆市 S6 安徽亳州 -
采用 Agilent C18(250 mm×4.6 mm, 5 μm)色谱柱;流动相为0.2%磷酸水溶液(A)-甲醇(B),梯度洗脱(0~5 min,90%→82.5% A;5~10 min,82.5%→75% A;10~30 min,75%→62% A;30~38 min,62%→58.3% A;38~43 min,58.3% A;43~48 min,58.3%→55% A;48~58 min,55→40% A;58~68 min,40%→20% A;68~72 min,20%→18.5% A;72~80 min,18.5%→5% A;80~90min,5%→90% A);流速:1 ml /min;柱温:室温;检测波长:220 nm;进样量:10 μl。
-
精密称取青蒿素、艾黄素、青蒿酸、猫眼草黄素、芹菜素、木犀草素、异绿原酸C、异绿原酸A、异绿原酸B、滨蒿内酯、东莨菪内酯对照品适量,用甲醇溶解得到浓度分别为300、31、96、78 、16、12、52、134、28、18、86 μg/ml的混合对照品溶液[11]。
-
药材粉末过50目筛后,精密称取1.5 g,加入甲醇50 ml,记录重量后超声(600 W,40 kHz,50 ℃)提取50 min,取出放置冷却至室温后加甲醇至之前的重量,混匀,过0. 45 μm微孔滤膜。
-
取“2.2.2”项下编号为S10的供试品溶液适量,按“2.1”项所述条件连续运行6次,计算得到RSD(色谱峰保留时间)均小于0.49%,RSD(峰面积)均小于1.93%,表明本研究所用设备精密度良好。
-
取“2.2.2”项下编号为S10的供试品溶液适量,分别于室温下放置0、2、4、8、12、16、24 h时按“2.1”项所述条件运行,计算得到RSD(色谱峰保留时间)均小于0.69%,RSD(峰面积)均小于4.67%,说明配制的溶液在1 d内具有稳定性。
-
取编号为S10的青蒿样品粉末1.5 g,共6份,精密称定,按“2.2.2”项和“2.1”项下制备供试品溶液并运行,计算得到色谱峰保留时间均小于0.72%,峰面积均小于4.76%,表明该方法重复性良好。
-
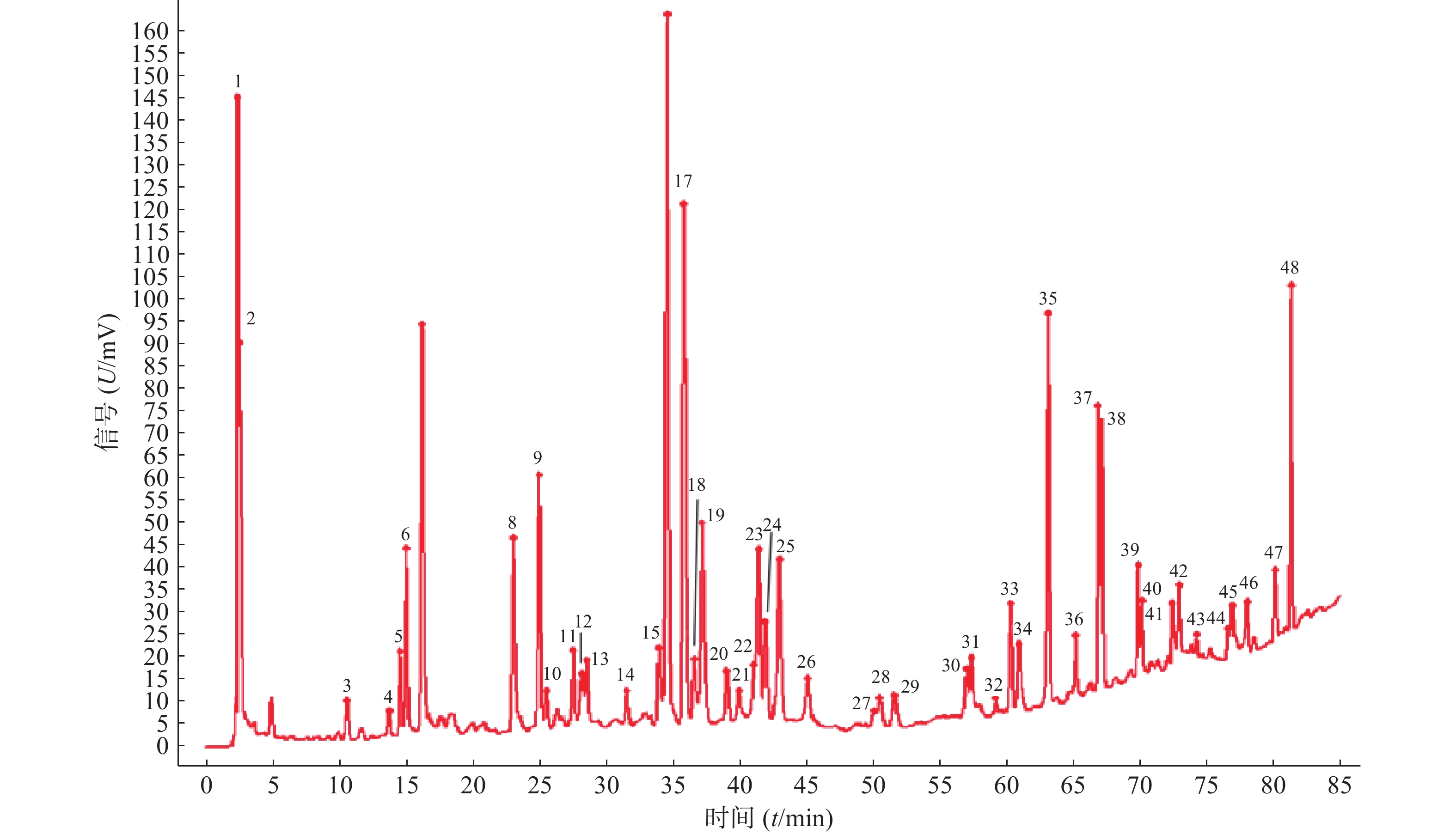
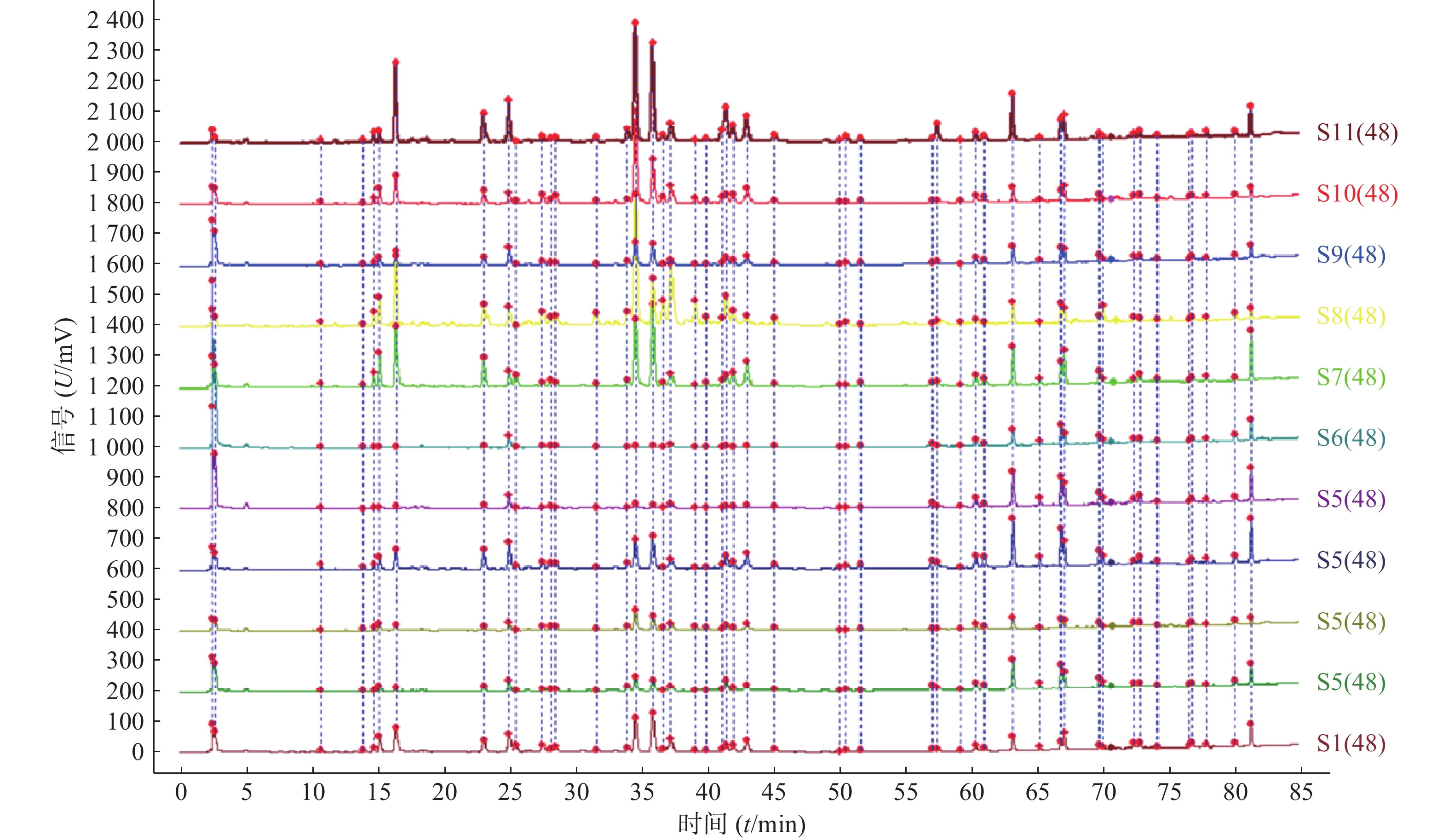
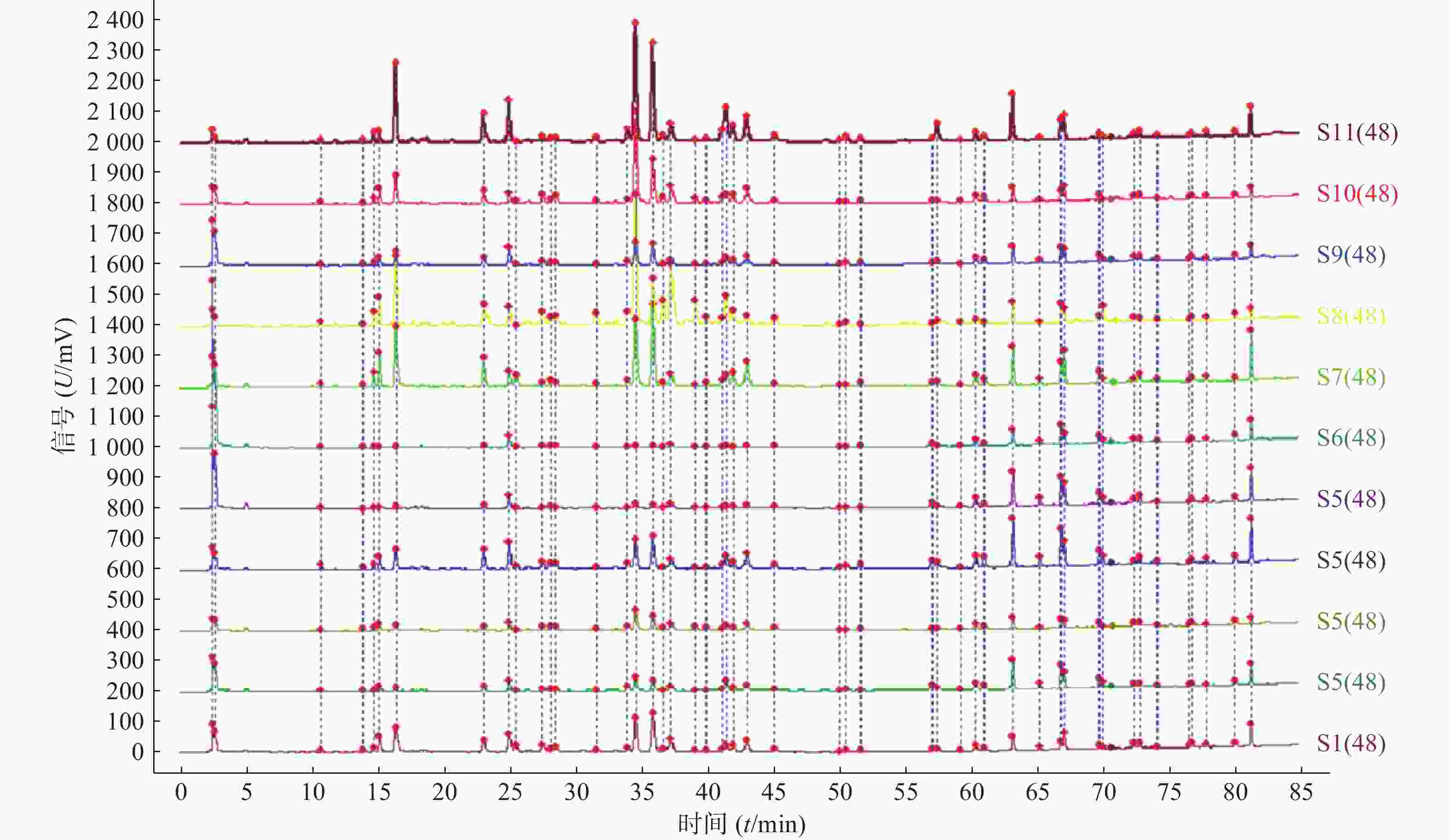
分别取11批青蒿样品,按照“2.2.2”项下方法制备供试品溶液,精密吸取供试品溶液10 μl,按“2.1”项下色谱条件进样检测后,将获得的HPLC数据导入《中药色谱指纹图谱相似度评价系统》(2012版),得到11批青蒿样品的指纹图谱,指认出48个共有峰,见图1、图2。
-
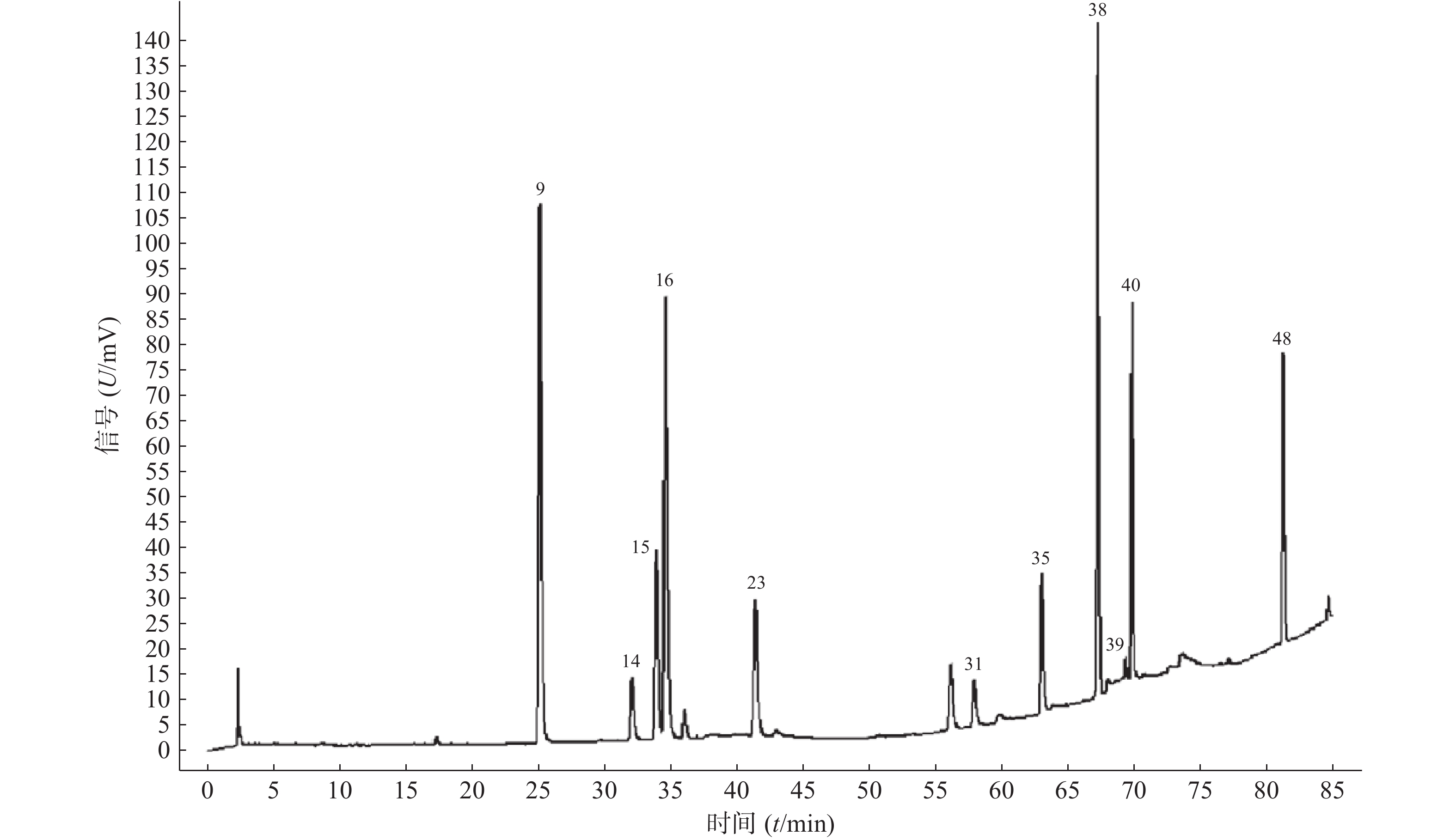
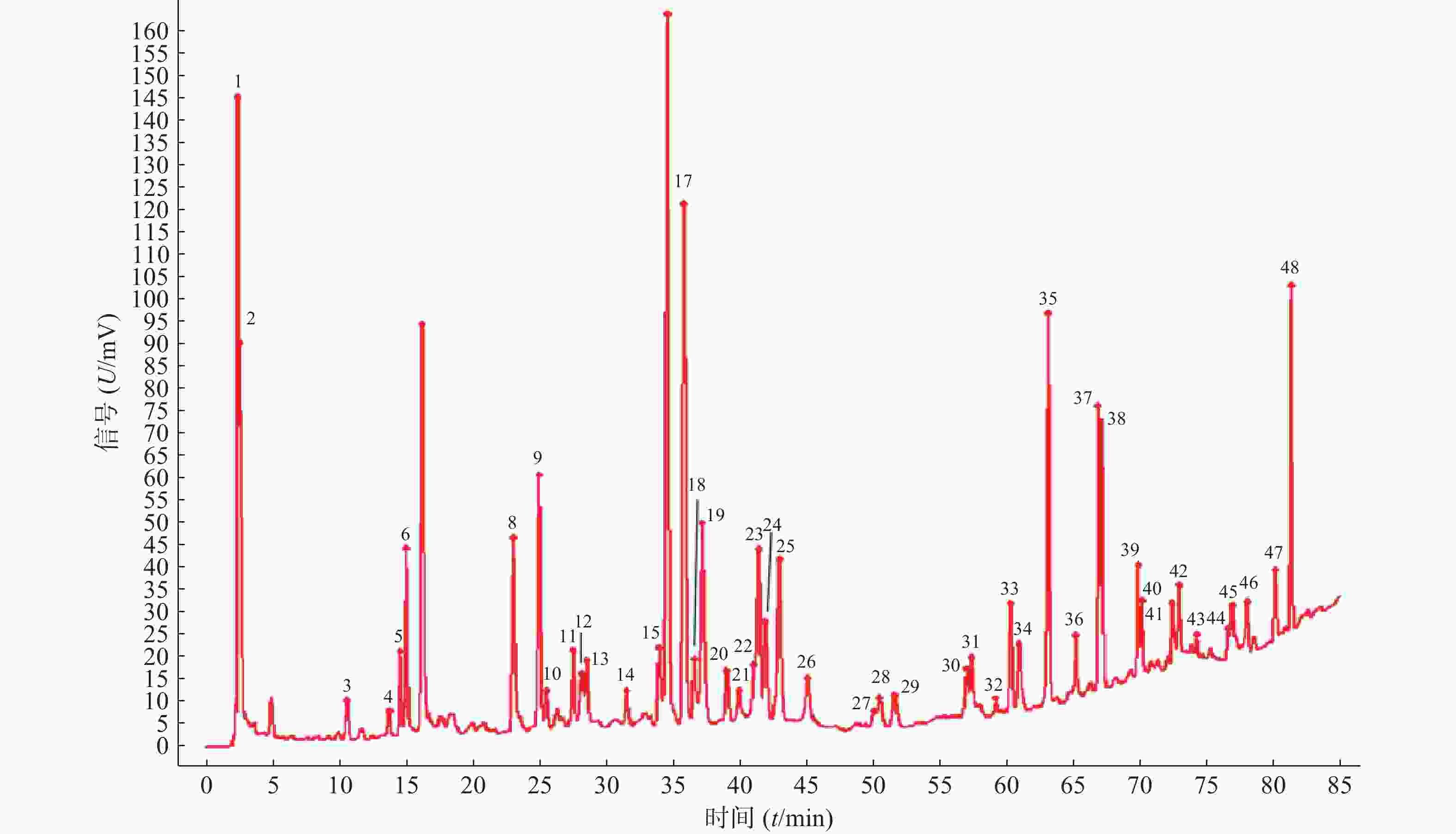
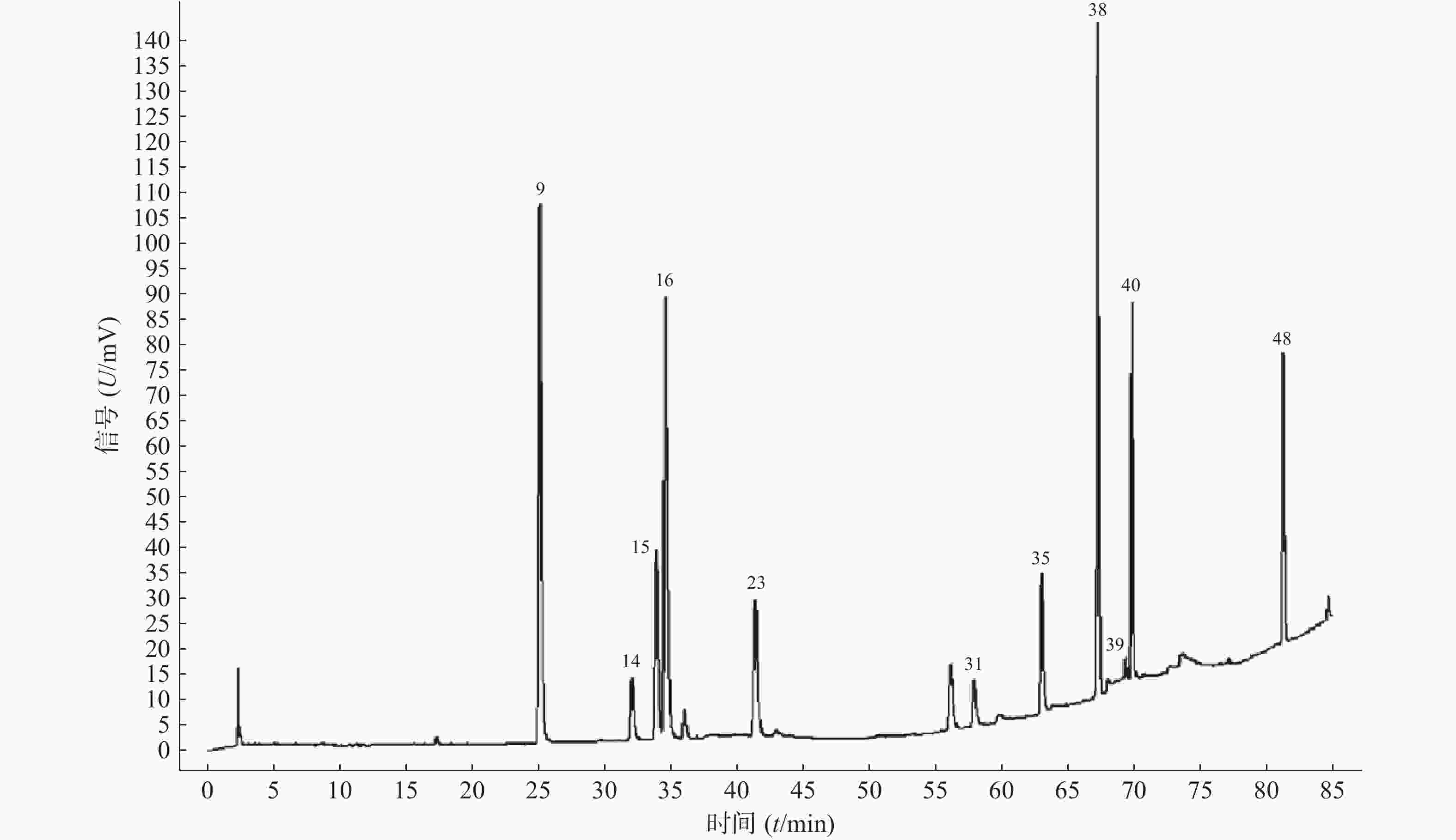
11批青蒿样品共有48个共有峰,共有峰占总峰比在0.776~0.864之间。选择S10为参照图谱,时间窗宽度设置为0.1 min,采用平均数法,以指纹图谱中峰形较好的色谱峰进行多点校正后生成叠加图谱,即为对照图谱。通过与混合对照品的HPLC分析结果对比,指认出9号峰为东莨菪内酯、14号峰为滨蒿内酯、15号峰为异绿原酸B、16号峰为异绿原酸A、23号峰为异绿原酸C、31号峰为木犀草素、35号峰为芹菜素、38号峰为猫眼草黄素、39号峰为青蒿素、40号峰为艾黄素、48号峰为青蒿酸(图3)。图谱显示,异绿原酸A峰面积大且稳定,分离度良好,故对比其保留时间(峰面积),其他峰的相对保留时间(峰面积)=其他峰的保留时间(峰面积)/异绿原酸A的保留时间(峰面积),见表2、表3。
编号 S1 S2 S3 S4 S5 S6 S7 S8 S9 S10 S11 RSD(%) 峰1 0.071 0.071 0.071 0.071 0.070 0.070 0.070 0.071 0.070 0.072 0.072 1.060 峰2 0.075 0.075 0.075 0.075 0.075 0.074 0.075 0.075 0.074 0.075 0.075 0.540 峰3 0.303 0.308 0.308 0.309 0.309 0.309 0.310 0.311 0.310 0.310 0.309 0.680 峰4 0.396 0.401 0.401 0.401 0.400 0.401 0.401 0.402 0.402 0.403 0.402 0.450 峰5 0.418 0.423 0.423 0.424 0.424 0.423 0.424 0.425 0.424 0.425 0.425 0.460 峰6 0.433 0.437 0.437 0.437 0.436 0.436 0.436 0.437 0.437 0.439 0.438 0.340 峰7 0.468 0.472 0.472 0.472 0.472 0.472 0.472 0.473 0.473 0.473 0.473 0.300 峰8 0.662 0.667 0.667 0.667 0.667 0.666 0.667 0.668 0.667 0.668 0.668 0.250 峰9 0.717 0.723 0.723 0.723 0.723 0.722 0.724 0.724 0.724 0.724 0.724 0.280 峰10 0.731 0.739 0.738 0.738 0.741 0.737 0.738 0.738 0.738 0.738 0.740 0.340 峰11 0.794 0.796 0.796 0.796 0.796 0.795 0.797 0.797 0.796 0.797 0.797 0.120 峰12 0.810 0.815 0.815 0.815 0.809 0.814 0.815 0.815 0.814 0.817 0.817 0.310 峰13 0.823 0.825 0.825 0.825 0.825 0.824 0.826 0.827 0.825 0.826 0.826 0.130 峰14 0.912 0.912 0.912 0.913 0.911 0.912 0.914 0.914 0.913 0.913 0.914 0.110 峰15 0.983 0.982 0.983 0.983 0.982 0.982 0.983 0.985 0.982 0.984 0.983 0.100 峰16 1.000 1.000 1.000 1.000 1.000 1.000 1.000 1.000 1.000 1.000 1.000 0 峰17 1.038 1.037 1.037 1.037 1.037 1.037 1.037 1.038 1.037 1.038 1.038 0.050 峰18 1.058 1.056 1.058 1.058 1.060 1.060 1.061 1.062 1.060 1.059 1.059 0.160 峰19 1.078 1.079 1.078 1.078 1.075 1.074 1.078 1.079 1.075 1.078 1.079 0.170 峰20 1.133 1.131 1.131 1.131 1.131 1.129 1.130 1.131 1.129 1.132 1.132 0.110 峰21 1.157 1.154 1.157 1.155 1.155 1.156 1.156 1.157 1.154 1.158 1.156 0.110 峰22 1.190 1.190 1.189 1.189 1.190 1.192 1.188 1.189 1.187 1.189 1.192 0.130 峰23 1.199 1.198 1.198 1.198 1.198 1.198 1.199 1.199 1.197 1.198 1.198 0.050 峰24 1.215 1.214 1.214 1.214 1.213 1.213 1.213 1.215 1.212 1.214 1.214 0.070 峰25 1.246 1.244 1.244 1.244 1.243 1.242 1.243 1.244 1.242 1.244 1.245 0.100 峰26 1.307 1.306 1.306 1.307 1.306 1.305 1.306 1.308 1.306 1.306 1.308 0.070 峰27 1.452 1.453 1.450 1.450 1.450 1.450 1.450 1.453 1.449 1.448 1.449 0.110 峰28 1.464 1.462 1.463 1.463 1.464 1.463 1.464 1.465 1.461 1.461 1.460 0.110 峰29 1.497 1.493 1.495 1.495 1.492 1.492 1.495 1.500 1.492 1.494 1.494 0.160 峰30 1.656 1.655 1.656 1.653 1.648 1.645 1.648 1.649 1.647 1.658 1.659 0.300 峰31 1.665 1.665 1.672 1.663 1.659 1.657 1.657 1.658 1.656 1.667 1.667 0.320 峰32 1.718 1.718 1.717 1.716 1.711 1.709 1.710 1.711 1.708 1.719 1.721 0.270 峰33 1.752 1.752 1.751 1.748 1.743 1.738 1.738 1.740 1.739 1.754 1.756 0.400 峰34 1.770 1.770 1.769 1.767 1.762 1.758 1.758 1.759 1.758 1.772 1.775 0.360 峰35 1.833 1.834 1.833 1.830 1.825 1.821 1.821 1.822 1.820 1.835 1.838 0.360 峰36 1.893 1.894 1.893 1.890 1.884 1.879 1.879 1.880 1.879 1.896 1.900 0.420 峰37 1.942 1.943 1.942 1.939 1.932 1.927 1.927 1.928 1.928 1.945 1.949 0.430 峰38 1.950 1.950 1.949 1.946 1.939 1.934 1.934 1.935 1.935 1.953 1.956 0.430 峰39 2.027 2.027 2.026 2.023 2.015 2.010 2.009 2.011 2.011 2.030 2.034 0.460 峰40 2.036 2.036 2.032 2.031 2.024 2.019 2.018 2.016 2.020 2.039 2.043 0.470 峰41 2.102 2.102 2.101 2.097 2.090 2.084 2.084 2.085 2.085 2.105 2.109 0.450 峰42 2.117 2.116 2.114 2.111 2.103 2.097 2.097 2.097 2.098 2.119 2.122 0.470 峰43 2.156 2.155 2.153 2.149 2.141 2.135 2.134 2.135 2.136 2.157 2.161 0.490 峰44 2.226 2.224 2.223 2.219 2.211 2.204 2.204 2.205 2.205 2.226 2.230 0.470 峰45 2.233 2.232 2.230 2.226 2.218 2.212 2.211 2.213 2.212 2.234 2.238 0.470 峰46 2.267 2.265 2.263 2.259 2.250 2.243 2.243 2.244 2.244 2.265 2.270 0.490 峰47 2.329 2.327 2.325 2.321 2.312 2.306 2.304 2.306 2.306 2.328 2.332 0.480 峰48 2.363 2.362 2.36 2.355 2.346 2.339 2.338 2.340 2.340 2.363 2.367 0.490 共有峰/总峰 0.837 0.815 0.778 0.821 0.811 0.834 0.864 0.776 0.823 0.806 0.838 编号 S1 S2 S3 S4 S5 S6 S7 S8 S9 S10 S11 RSD(%) 峰1 0.221 0.718 0.167 0.215 6.001 18.615 0.134 0.037 0.545 0.046 0.033 232.240 峰2 0.539 2.042 0.539 0.505 11.735 36.124 0.337 0.055 1.571 0.192 0.041 223.380 峰3 0.065 0.102 0.060 0.131 0.300 0.356 0.050 0.035 0.070 0.032 0.028 100.170 峰4 0.066 0.061 0.059 0.088 0.121 0.476 0.033 0.015 0.064 0.021 0.026 139.490 峰5 0.134 0.056 0.104 0.130 0.183 0.339 0.145 0.084 0.080 0.054 0.073 64.820 峰6 0.391 0.338 0.288 0.344 0.243 0.408 0.321 0.169 0.271 0.131 0.077 39.430 峰7 0.987 0.201 0.214 0.514 0.385 0.315 0.850 0.405 0.308 0.251 0.482 57.330 峰8 0.375 0.295 0.217 0.584 0.686 0.903 0.376 0.150 0.338 0.143 0.305 59.310 峰9 0.533 0.646 0.316 0.766 2.647 4.610 0.201 0.130 0.674 0.112 0.332 139.510 峰10 0.223 0.058 0.024 0.137 0.259 0.464 0.189 0.008 0.051 0.039 0.023 104.800 峰11 0.225 0.203 0.202 0.236 0.365 0.952 0.079 0.120 0.145 0.112 0.069 101.160 峰12 0.119 0.213 0.234 0.245 0.516 0.872 0.116 0.072 0.117 0.055 0.049 105.050 峰13 0.197 0.181 0.207 0.214 0.376 1.106 0.085 0.107 0.143 0.111 0.061 116.600 峰14 0.055 0.151 0.081 0.096 0.132 0.242 0.064 0.129 0.067 0.047 0.058 57.120 峰15 0.125 0.315 0.186 0.226 0.295 0.477 0.109 0.138 0.179 0.062 0.126 58.660 峰16 1.000 1.000 1.000 1.000 1.000 1.000 1.000 1.000 1.000 1.000 1.000 0 峰17 1.130 0.724 0.667 1.118 0.879 0.891 1.175 0.356 0.943 0.466 0.849 31.770 峰18 0.104 0.101 0.185 0.115 0.241 0.060 0.089 0.224 0.053 0.108 0.087 51.150 峰19 0.551 0.465 0.406 0.497 1.441 1.610 0.265 0.566 0.291 0.253 0.229 79.600 峰20 0.050 0.093 0.222 0.077 0.080 0.171 0.041 0.214 0.047 0.079 0.029 69.220 峰21 0.086 0.169 0.206 0.138 0.095 0.248 0.085 0.079 0.120 0.066 0.058 50.140 峰22 0.096 0.098 0.081 0.120 0.130 0.177 0.105 0.050 0.112 0.069 0.078 33.720 峰23 0.251 0.842 0.321 0.530 0.626 0.594 0.206 0.256 0.418 0.121 0.353 52.610 峰24 0.237 0.280 0.147 0.286 0.324 0.264 0.244 0.138 0.289 0.122 0.176 30.790 峰25 0.445 0.565 0.441 0.730 0.725 0.619 0.486 0.121 0.558 0.243 0.306 40.560 峰26 0.175 0.211 0.243 0.279 0.427 0.832 0.121 0.107 0.199 0.093 0.120 83.960 峰27 0.050 0.045 0.030 0.090 0.161 0.245 0.056 0.006 0.071 0.028 0.027 95.860 峰28 0.063 0.243 0.054 0.177 0.170 0.123 0.049 0.036 0.127 0.018 0.080 68.020 峰29 0.107 0.125 0.092 0.177 0.361 0.428 0.091 0.015 0.109 0.053 0.041 90.430 峰30 0.080 0.390 0.063 0.298 1.373 1.312 0.065 0.019 0.113 0.026 0.030 148.400 峰31 0.071 0.216 0.066 0.163 0.244 0.216 0.065 0.023 0.100 0.030 0.158 64.710 峰32 0.027 0.071 0.019 0.054 0.303 0.210 0.030 0.014 0.032 0.017 0.018 131.430 峰33 0.160 0.445 0.234 0.380 1.852 2.801 0.146 0.036 0.261 0.087 0.075 151.790 峰34 0.114 0.280 0.166 0.312 0.777 1.295 0.117 0.019 0.159 0.054 0.055 128.230 峰35 0.336 1.630 0.399 1.267 6.102 5.469 0.423 0.117 0.603 0.123 0.317 141.670 峰36 0.070 0.330 0.042 0.260 1.421 1.875 0.063 0.004 0.116 0.013 0.021 167.530 峰37 0.153 0.999 0.269 0.774 3.799 5.304 0.195 0.088 0.399 0.071 0.106 159.420 峰38 0.304 0.702 0.221 0.534 3.139 3.099 0.303 0.068 0.400 0.101 0.143 140.610 峰39 0.090 0.447 0.148 0.331 1.801 2.513 0.123 0.022 0.194 0.042 0.033 159.710 峰40 0.084 0.285 0.091 0.199 1.102 1.365 0.043 0.076 0.122 0.021 0.016 151.000 峰41 0.190 0.366 0.232 0.172 1.131 2.101 0.068 0.033 0.226 0.055 0.039 152.190 峰42 0.157 0.322 0.185 0.229 1.672 2.273 0.121 0.028 0.231 0.047 0.052 155.930 峰43 0.065 0.137 0.091 0.109 0.535 1.037 0.049 0.017 0.093 0.027 0.027 157.670 峰44 0.059 0.108 0.068 0.049 0.303 0.650 0.019 0.012 0.062 0.017 0.014 156.080 峰45 0.110 0.234 0.138 0.113 0.747 1.344 0.045 0.020 0.135 0.033 0.025 153.940 峰46 0.083 0.114 0.089 0.161 0.705 1.559 0.064 0.024 0.127 0.032 0.049 170.940 峰47 0.108 0.285 0.176 0.201 1.054 2.412 0.039 0.039 0.161 0.037 0.038 175.050 峰48 0.389 0.878 0.171 0.854 4.399 5.353 0.419 0.045 0.356 0.065 0.162 155.940 -
将试剂盒中Trolox溶液按说明书稀释成梯度浓度,加入反应液、不同浓度的Trolox溶液,并设置空白对照。反应结束后,在相应波长下测出测定孔吸光度Ac、空白孔吸光度Ak,自由基清除率(%)=(1−(Ac−Ak)÷Ak)×100%,以浓度(Y)对DPPH自由基清除率(X)进行线性回归,获得标准回归曲线方程,见表4,以标准回归曲线方程计算得出自由基清除能力。
自由基 标准回归曲线方程 r DPPH Y=0.021 8X+0.005 7 0.999 9 ABTS Y=0.447 6X+0.012 4 0.997 3 -
取11批供试品溶液(编号:S1~11,按“2.2.2”项下方法制备)各1 ml,加无水甲醇9 ml稀释10倍;对照孔加入供试品100 μl、无水甲醇150 μl,测定孔加入供试品100 μl、DPPH工作液150 μl,空白孔加入供试品100 μl、DPPH工作液150 μl后混匀,避光室温下静置0.5 h,于540 nm波长处测得对照孔吸光度Ad,测定孔吸光度Ac,空白孔吸光度Ak,DPPH自由基清除率(%)=(1−(Ac-Ad)÷Ak)×100%,供试品DPPH自由基清除能力(mmol/L Trolox)=带入标准回归曲线方程所得Trolox的浓度×10,结果见表5。11批青蒿样品中,样品8(编号:S8)清除DPPH自由基的能力最强。
编号 DPPH清除能力 ABTS清除能力 S1 2.725 6.243 S2 1.336 2.025 S3 2.765 4.521 S4 3.110 7.190 S5 1.617 2.848 S6 0.970 0.853 S7 5.057 8.641 S8 6.477 10.453 S9 2.025 2.575 S10 4.731 8.313 S11 5.321 9.848 -
取11批供试品溶液(编号:S1~11,按“2.2.2”项下方法制备)各1 ml,加无水甲醇9 ml稀释10倍;对照孔加入供试品100 μl、无水甲醇150 μl,测定孔加入供试品100 μl、ABTS工作液150 μl,空白孔加入无水乙醇100 μl、ABTS工作液150 μl,混匀置于室温反应6 min,于405 nm波长处测得对照孔吸光度Ad,测定孔吸光度Ac,空白孔吸光度Ak,DPPH自由基清除率(%)=(1−(Ac−Ad)÷Ak)×100%,供试品ABTS自由基清除能力(mmol/L Trolox)=代入标准回归曲线方程所得Trolox的浓度×10,结果见表5。11批青蒿样品中,样品8(编号:S8)清除ABTS自由基的能力最强。
-
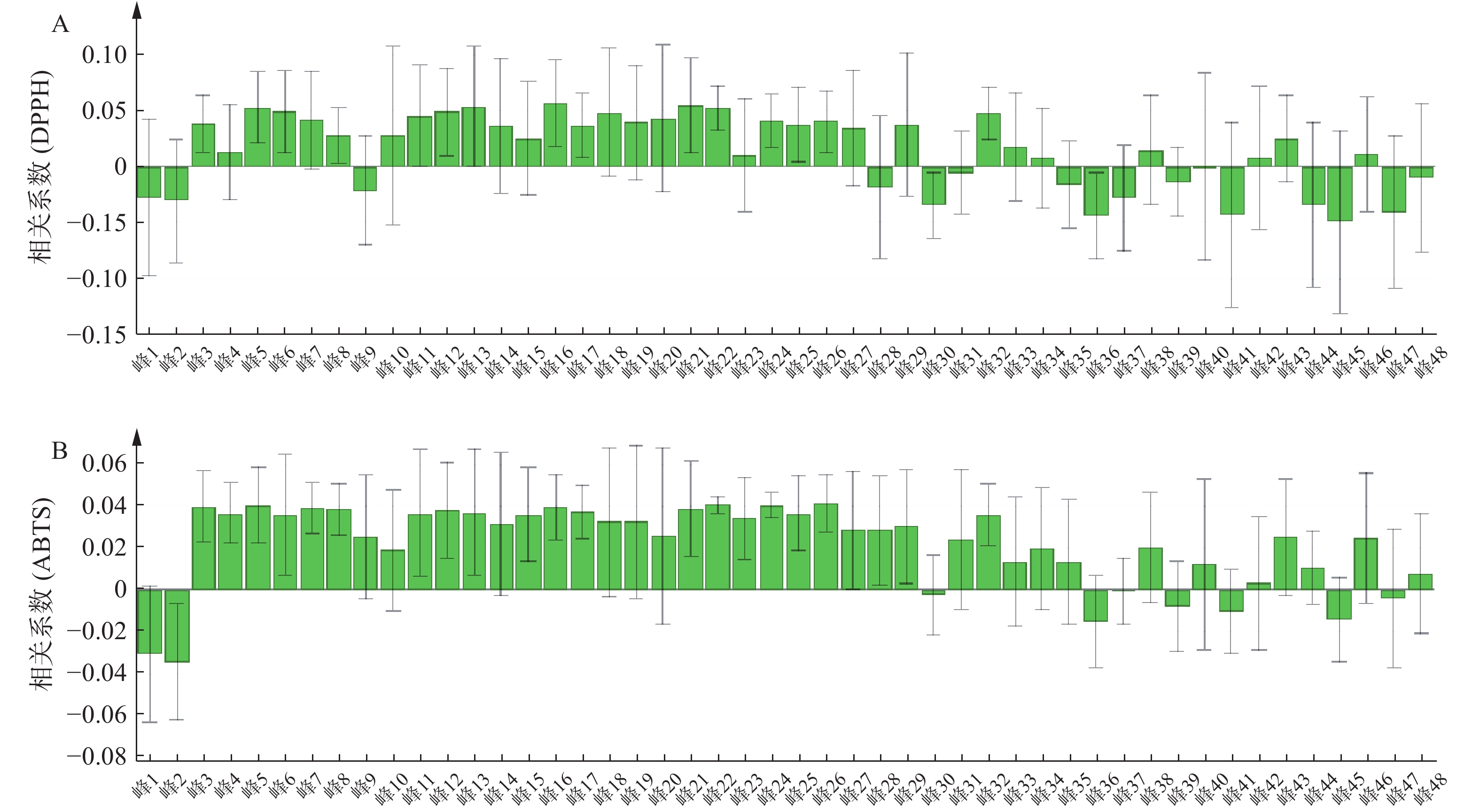
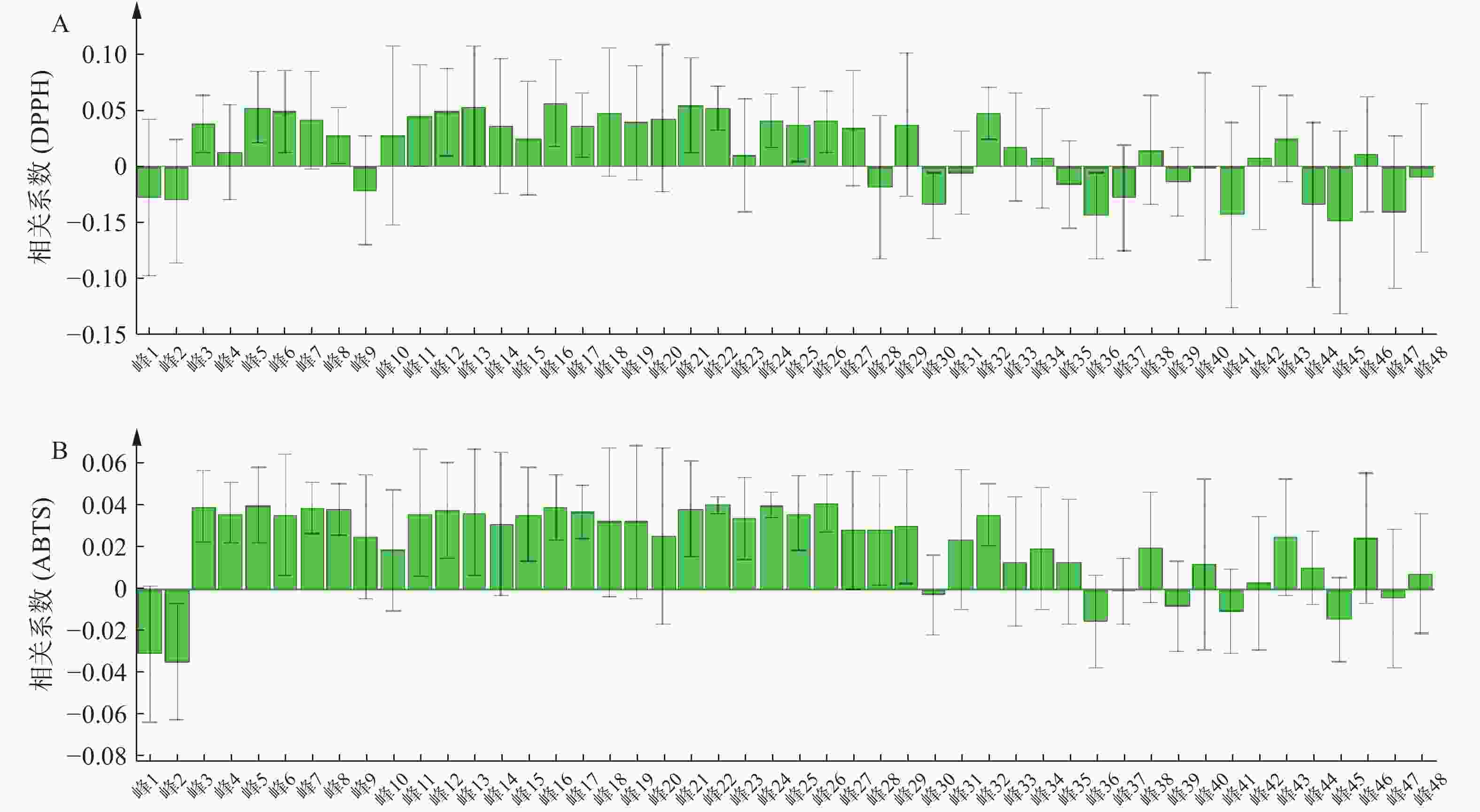
将表5青蒿抗氧化活性结果和表3的相对峰面积数据导入SIMCA 14.1软件构建PLSR模型。结果显示,峰3~峰8、峰10~峰27、峰29、峰32~峰34、峰38、峰40、峰42~峰43、峰46与DPPH自由基清除率呈正相关;峰3~峰29、峰31~峰35、峰38、峰40、峰42~峰44、峰46、峰48与ABTS自由基清除率呈正相关,结果见图4。
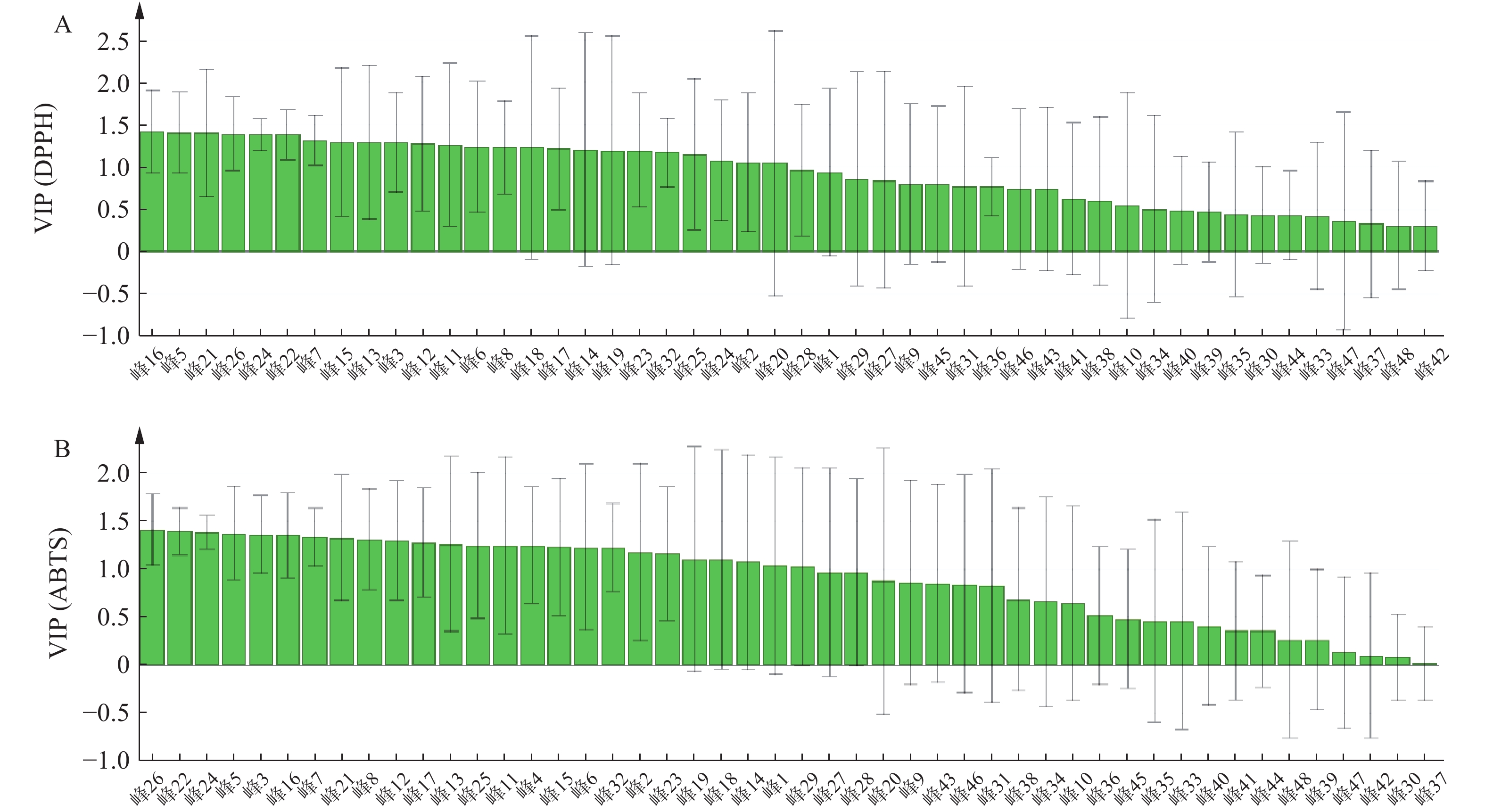
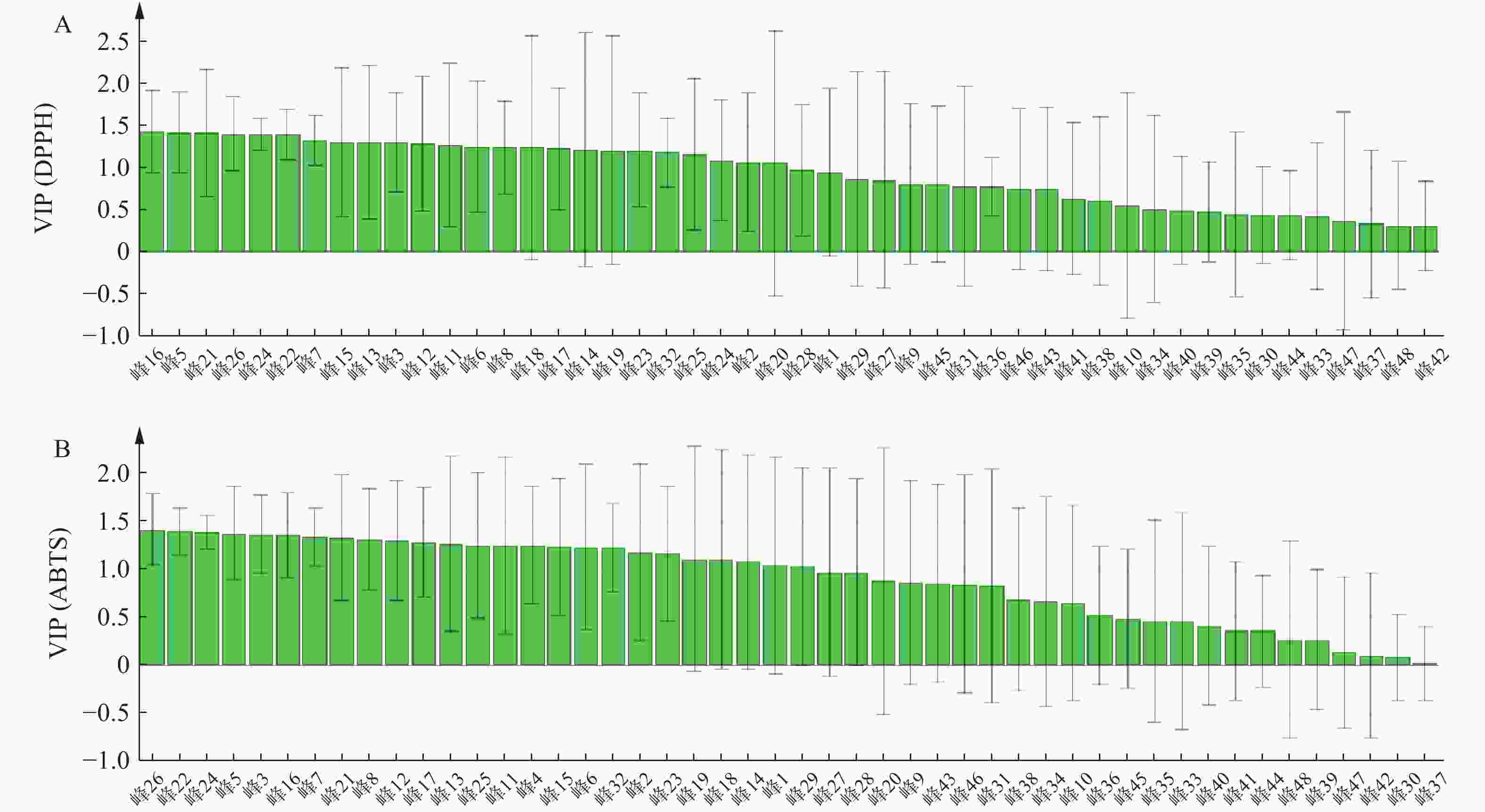
变量投影(VIP)用于评估每个自变量对因变量的重要性,VIP>1表明自变量对因变量的贡献有显著性。筛选出对自由基清除能力呈正相关,且VIP>1的峰。结果显示,青蒿药材样品已明确成分中对DPPH自由基清除能力的贡献大小依次为异绿原酸A(16号峰)>异绿原酸B(15号峰)>滨蒿内酯(14号峰)>异绿原酸C(23号峰);青蒿药材样品已明确成分中对ABTS自由基清除能力的贡献大小依次为异绿原酸A(16号峰)>异绿原酸B(15号峰)>异绿原酸C(23号峰)>滨蒿内酯(14号峰),提示相应成分与青蒿抗氧化能力呈正相关,结果见图5。
-
利用HPLC法测定青蒿药材样品的图谱,能准确、稳定地检测青蒿中的多种有效成分。实验前期研究发现,在乙腈-0.1%磷酸水、甲醇-0.1%磷酸水等流动相中,甲醇-0.1%磷酸水为流动相的分离效果最优,但青蒿素和艾黄素的分离度不佳;随后将水相改为0.2%磷酸水后,分离度得到了提高。研究前期考察了多个波长下各成分的显峰情况,发现在高波长下青蒿素不显峰,故最后选择220 nm为指纹图谱波长,但低波长时显峰较多,在一定程度上影响了几种已知成分的VIP值。
本研究以11批不同地区青蒿为研究对象,采用HPLC法研究其各共有峰与体外抗氧化能力之间的关系,探究青蒿体外抗氧化作用的药效物质基础。结果显示,异绿原酸A、异绿原酸B、异绿原酸C和滨蒿内酯与自由基清除率呈正相关,提示这些成分是青蒿体外抗氧化的药效物质基础。青蒿素是青蒿中的重要成分,有文献研究发现其具有抗氧化的作用[12],也有文献研究发现其可以激活氧化应激[13],而本研究发现青蒿素的抗氧化作用并不明显。推测原因可能在于本研究所用的是基于清除自由基的抗氧化测试方法,青蒿素可能是通过清除自由基之外的途径起到抗氧化作用[14],仍有待今后深入研究。
Study on spectrum-effect relationship based on antioxidant activity of Artemisiae Annuae Herba
doi: 10.12206/j.issn.2097-2024.202211012
- Received Date: 2022-11-07
- Rev Recd Date: 2024-01-26
- Available Online: 2024-05-22
- Publish Date: 2024-05-25
-
Key words:
- Artemisiae Annuae Herba /
- HPLC /
- antioxidant /
- spectrum-effect relationship /
- pharmacodynamic material basis
Abstract:
| Citation: | LAI Liyong, XIA Tianshuang, XU Shengyan, JIANG Yiping, YUE Xiaoqiang, XIN Hailiang. Study on spectrum-effect relationship based on antioxidant activity of Artemisiae Annuae Herba[J]. Journal of Pharmaceutical Practice and Service, 2024, 42(5): 203-210, 216. doi: 10.12206/j.issn.2097-2024.202211012 |







 DownLoad:
DownLoad: