-
近年来,我国海军大力加强实战化军事训练,不断提高军事应对能力和应急突击能力,高新技术条件下的未来海上作战对药材保障的要求越来越高,药材保障是影响救治的主要因素之一[1]。海上高温、高湿、高盐环境对药品的运输、储存和使用提出了新的考验。磺胺嘧啶银乳膏为磺胺类抗菌药,临床常用于预防和治疗轻度烧伤继发创面感染等,是一种疗效好、应用广泛的外用抗菌药物。药物制剂的含量符合规定标准是保障药物疗效的重要指标之一。目前,磺胺嘧啶银乳膏含量测定的方法多种多样,《中国药典(二部)》2015年版采用的是永停滴定法[2],《中国医院制剂规范》(第二版)则采用硫氰酸铵滴定法[3],而《美国药典》采用HPLC法测定磺胺嘧啶银的含量[4]。用滴定法测定含量时容易受到基质的干扰,影响终点的判断,《美国药典》方法的供试品前处理烦琐。根据已报道的文献资料[5-6],本研究对磺胺嘧啶银乳膏含量测定方法进行优化,建立了反相HPLC测定磺胺嘧啶银乳膏中该成分含量的方法,并首次采用复合式盐雾试验箱模拟海洋高温、高湿、高盐环境,考察了磺胺嘧啶银乳膏在极端环境下的稳定性,为特殊环境下的药品质量研究提供方法借鉴,并为优化远海作战药材保障提供数据支持。
HTML
-
LC-20A高效液相色谱仪,包括LC-20AT紫外检测器和Labsolution工作站(日本岛津公司) METTLER AE240型电子天平(瑞士梅特勒公司),ZH-AYSH-90复合式盐雾试验箱 (香港正航仪器设备有限公司)。
-
对照品:磺胺嘧啶银(USP,批号:RO3500,纯度99.9%),磺胺嘧啶银乳膏(海军军医大学附属长海医院,规格:1%,批号:20191207、20191208、20191209),乙腈为色谱纯,氨水为分析纯,磷酸为优级纯(国药集团化学试剂有限公司),实验用水为娃哈哈纯净水。
1.1. 仪器
1.2. 试药
-
色谱柱:Agilent ZORBAX SB-C18 (4.6 mm×150 mm,5μm);流动相:乙腈-0.1%磷酸 (8∶92,V/V),流速1.0 ml/min,进样量10 μl,柱温30℃,检测波长254 nm。
-
取磺胺嘧啶银对照品10 mg,精密称定,置50 ml棕色量瓶中,加入10%氨水溶液溶解稀释至刻度,摇匀,作为对照品储备液(0.2 mg/ml),精密量取对照品储备液0.5 ml置10 ml量瓶中,加10%氨水溶液稀释至刻度,摇匀,即得磺胺嘧啶银对照品溶液。
-
将磺胺嘧啶银乳膏置于复合式盐雾试验箱中,温度50℃,湿度90%,盐度5%氯化钠[7-8],分别放置0、2、4、6、8、10、12周取样测定。
精密称定磺胺嘧啶银乳膏1 g,置于50 ml棕色量瓶中,加入10%氨水溶液适量,超声20 min,使乳膏充分溶解并稀释至刻度,过滤,取续滤液0.5 ml,置于10 ml量瓶,用流动相稀释至刻度,摇匀,即为供试品溶液。
-
根据磺胺嘧啶银乳膏处方制备不含磺胺嘧啶银的空白乳膏剂,按照“2.3”项下制备方法制成阴性样品溶液。
-
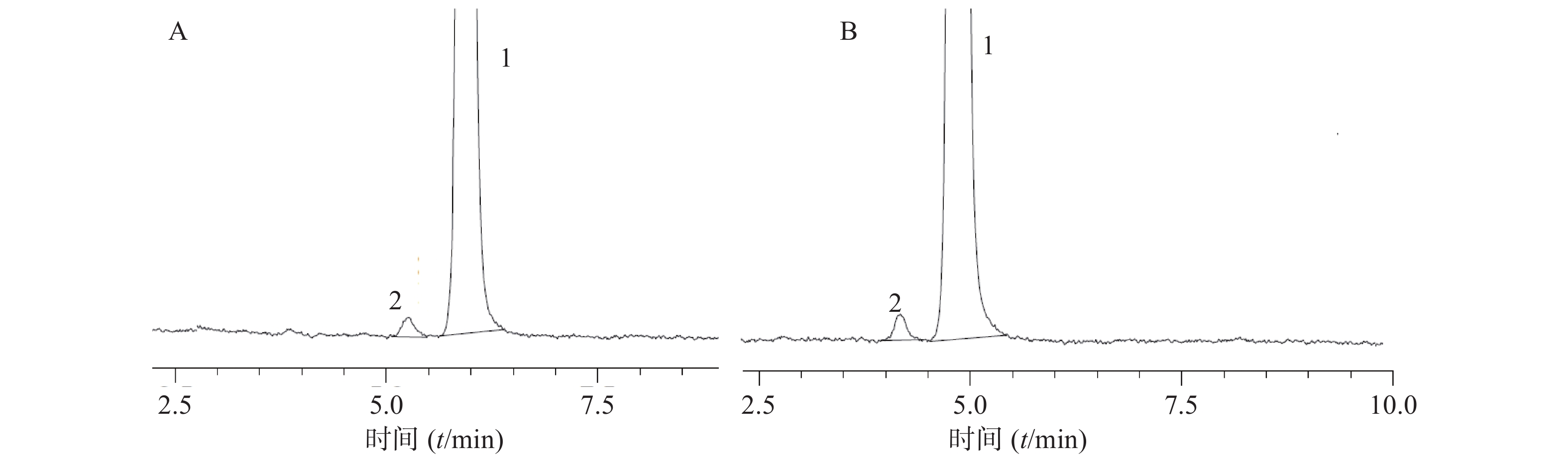
分别取阴性样品溶液、对照品溶液及供试品溶液各10 μl,按“2.1”项下的色谱条件进样分析,结果如图1所示,供试品溶液在与对照品溶液对应位置出现相对应的色谱峰,阴性样品溶液无干扰,方法专属性良好。
-
精密量取对照品储备液适量置于10 ml棕色量瓶中,用10%氨水稀释,依次配制成浓度分别为4.00、8.00、10.00、12.00、16.00、20.00 μg/ml的对照品溶液,作为线性工作溶液。按“2.1”项下色谱条件,进样10 μl,以磺胺嘧啶银的浓度为横坐标(X),峰面积为纵坐标(Y),进行线性回归,得到回归方程:Y=22 808X−1 858,r =0.999 9,结果表明,磺胺嘧啶银在4.00~20.00 μg/ml范围内与峰面积呈良好的线性关系。
-
取对照品溶液适量,按“2.1”项下色谱条件在1 d内进样6次,以及连续3d分别进样,测定磺胺嘧啶银峰面积,结果显示,日内精密度RSD为0.33%,日间精密度RSD为0.97%,表明仪器的精密度良好。
-
取同一批号(20191208)样品1 g,共6份,精密称定,按照“2.3”项下方法制备供试品溶液,按“2.1”项下色谱条件分别测定,结果显示,磺胺嘧啶银平均含量为95.27%,RSD为1.07% (n=6),表明方法的重复性良好。
-
取同一份供试品溶液,室温下分别放置0、2、4、8、12、24 h后分别按“2.1”项下色谱条件进样分析,考察溶液的稳定性,测定结果的RSD为0.97%,表明供试品溶液至少在24 h内稳定性良好。
-
精密称取磺胺嘧啶银乳膏空白基质6份,各1 g,置于50 ml量瓶中,分别加入磺胺嘧啶银对照品10 mg,再加入10%氨水溶液适量,超声后按“2.3”项下方法进行制备并测定,计算回收率,结果平均回收率为101.47%,RSD为2.33% (n=6)。
-
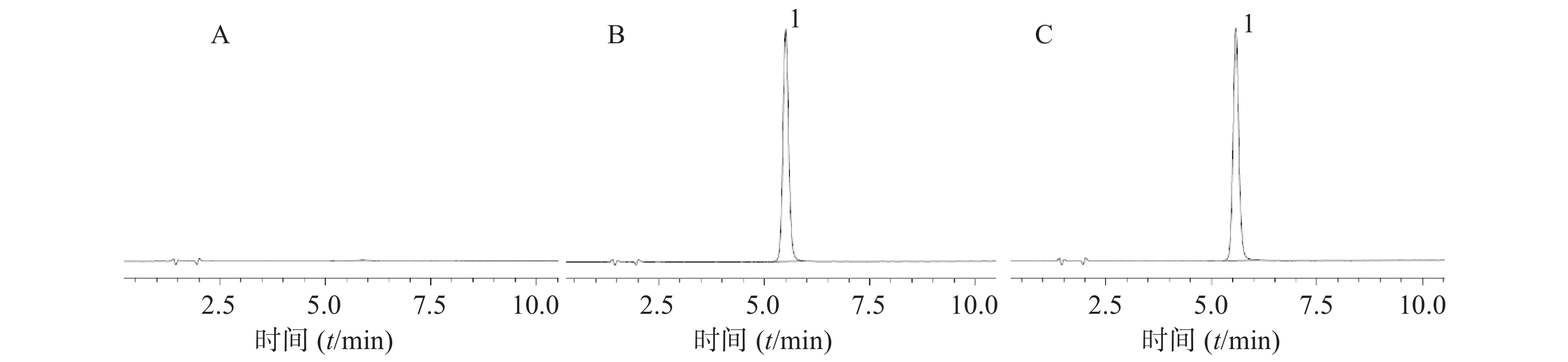
考察包括色谱柱(Agilent ZORBAX SB-C18: 4.6 mm×150 mm,5 μm,Agilent EXTEND-C18:4.6 mm×250 mm,5 μm),柱温(28、30、32℃),流速(0.9、1.0、1.1ml/min)3个因素对试验结果的影响,结果显示,磺胺嘧啶银峰面积的 RSD 均<10%,其中,微调流速对保留时间和峰面积的影响较大,但主成分与降解后出现的新杂质分离度良好,不影响含量测定结果,如图2所示,表明该方法具有良好的耐用性。
-
分别取3个批次的样品,按照“2.3”项下方法制备供试品溶液,用“2.1”项下色谱条件测定,记录峰面积,按外标法分别计算磺胺嘧啶银的含量,结果如表1所示。
批号 存储时间(t/w) 0 2 4 6 8 10 12 20191207 94.22 93.40 94.07 85.28 57.86 63.23 52.33 20191208 94.63 94.09 94.10 82.94 60.19 58.25 51.41 20191209 95.47 96.75 95.93 78.74 66.58 53.25 51.89
2.1. 色谱条件
2.2. 对照品溶液的制备
2.3. 供试品溶液的制备
2.4. 阴性样品溶液的制备
2.5. 专属性试验
2.6. 线性及范围
2.7. 精密度试验
2.8. 重复性试验
2.9. 稳定性试验
2.10. 回收率试验
2.11. 耐用性考察
2.12. 含量测定
-
磺胺嘧啶银是难溶的两性药物,只能在硝酸、氨水等少数溶剂中溶解[9]。本研究采用10%氨水溶解磺胺嘧啶银,并用50℃超声处理,使基质分散,冷却至室温后,定容,静置20 min,过滤,再用流动相稀释,HPLC测定磺胺嘧啶银的含量,并进行了完整的方法学考察,符合药典规定的各项要求。与《美国药典》的样品前处理方法相比,该方法操作简单、容易过滤、重复性良好。
乳膏剂常规应保存在阴凉干燥处,然而,远海作战环境特殊,高温、高湿、高盐是其主要特征,例如,担负海上救护任务的医院船,船舱多为密闭空间,舱内温度高,不利于药品的长期保存[10]。在如此特殊的环境下,药品是否能在其拟定的保质期内保持质量的稳定性,如何确定或延长药品在极端环境下的保质期是值得深入研究的课题。本研究首次采用复合式盐雾试验箱模拟高温、高湿、高盐环境,考察磺胺嘧啶银乳膏的稳定性,结果显示,在此极端环境下,磺胺嘧啶银乳膏在1个月内,其性状、pH值、粒度和含量没有发生显著变化,2个月以后,乳膏由白色转为褐色,明显呈水样状,有较大的块状凝结,含量下降超过30%,结果表明磺胺嘧啶银乳膏在温高、湿高、盐环境下不稳定,在其运输、储存和使用过程中应充分考虑环境的影响,对于长远航任务应考虑对该制剂的外包装加以防护措施,或更换同类型的其他品种。本研究可为极端环境下的药品稳定性研究方法的开发和运用提供参考。








 DownLoad:
DownLoad: