-
盐酸安罗替尼(AL3818)商品名福可维,是我国自主研发的1.1类新药。安罗替尼是一种以血管内皮生长因子受体(VEGFR)、成纤维细胞生长因子受体(FGFR)、血小板衍生生长因子(PDGFR)、干细胞因子(c-kit)受体为靶点的新型口服多靶点酪氨酸激酶抑制剂(TKIs)[1]。截止目前安罗替尼已获国家药品监督管理局(NMPA)批准,用于三线治疗非小细胞肺癌(NSCLC)和小细胞肺癌(SCLC),以及二线治疗软组织肉瘤(STS)。同时,安罗替尼单药或联合放化疗、PD-1/PDL-1治疗恶性肿瘤的多个临床试验正在开展中。预计会有越来越多的患者使用安罗替尼,因此亟需了解其不良反应特性。
与大部分VEGFR抑制剂相同,高血压也是安罗替尼常见的不良反应之一,然而,在临床试验中高血压发生率报道具有很大的差异。先前的荟萃分析(Meta-analysis)表明,使用VEGFR抑制剂与高血压发生风险增加相关[2-7],但目前国内外暂无关于安罗替尼高血压发生率和发生风险的Meta分析报道。由于高血压控制不佳可能导致剂量下调、严重的心血管事件甚至危及生命的后果,所以确定与安罗替尼相关的高血压发生率和发生风险十分重要。本研究拟对已发表的临床试验进行Meta分析,以确定其总体发生率和风险。同时探索VEGFR抑制剂之间高血压发生的差异,这可能会为理解其潜在机制以及制定治疗策略提供更多的参考依据。
HTML
-
英文以“anlotinib”“AL3818”“clinical trial”等为检索词,检索Pubmed、Cochrane Library、Embase数据库,同时检索美国临床肿瘤学会(ASCO)获得会议论文,通过ClinicalTrials.gov网站获得相关临床试验信息。中文以“安罗替尼”“福可维”“AL3818”“临床试验”等为检索词,检索中国知网、万方、维普和中国生物医学文献服务系统数据库。检索时间为建库至2020年7月,收集国内外公开发表的与安罗替尼相关的临床试验。
-
纳入标准:①前瞻性Ⅱ期和Ⅲ期临床试验;②受试者经病理组织学或细胞学检查证实为癌症患者;③试验组使用安罗替尼单药;④结局指标至少包含以下其中一种:发生所有等级或高等级(≥3级)高血压事件。
排除标准:①重复发表的文献;②动物实验;③Ⅰ期临床试验;④回顾性研究;⑤观察性研究;⑥病例报道;⑦试验组联合用药;⑧未详细报道本研究定义的结局指标;⑨通讯或评论;⑩同一试验的亚组分析。
-
由2名研究者独立进行文献筛选和数据提取,并进行交叉核对,如出现分歧则通过讨论或咨询第三方解决。对于每一项研究提取以下信息:第一作者姓名、恶性肿瘤种类、试验阶段和类型、治疗方案和对照方案、纳入分析的患者人数及总人数以及发生高血压事件的人数等。纳入的研究根据美国国家癌症研究所的统一评价标准(3.0或4.0版本;http://ctep.cancer.gov)对高血压事件进行评估和记录,高等级(≥3级)为严重的不良事件。
-
采用R软件(3.6.0版本)中的meta程序包进行Meta分析,根据提取到的数据,计算与安罗替尼相关的高血压发生率及其95%置信区间(CI)。为了准确计算相对风险(RR)及其95% CI,仅纳入对照组为安慰剂的随机对照试验(RCT)的数据。采用I2和Q统计量评估纳入研究的异质性,I2≥50%,P<0.05表明异质性较大,采用随机效应模型进行合并分析,反之,使用固定效应模型进行合并分析。应用SPSS软件(26.0版本),分析比较安罗替尼与各种VEGFR抑制剂的高血压发生率。双侧P<0.05表示差异具有统计学意义。
1.1. 检索策略
1.2. 文献纳入和排除标准
1.3. 数据提取和临床结局指标
1.4. 统计学分析
-
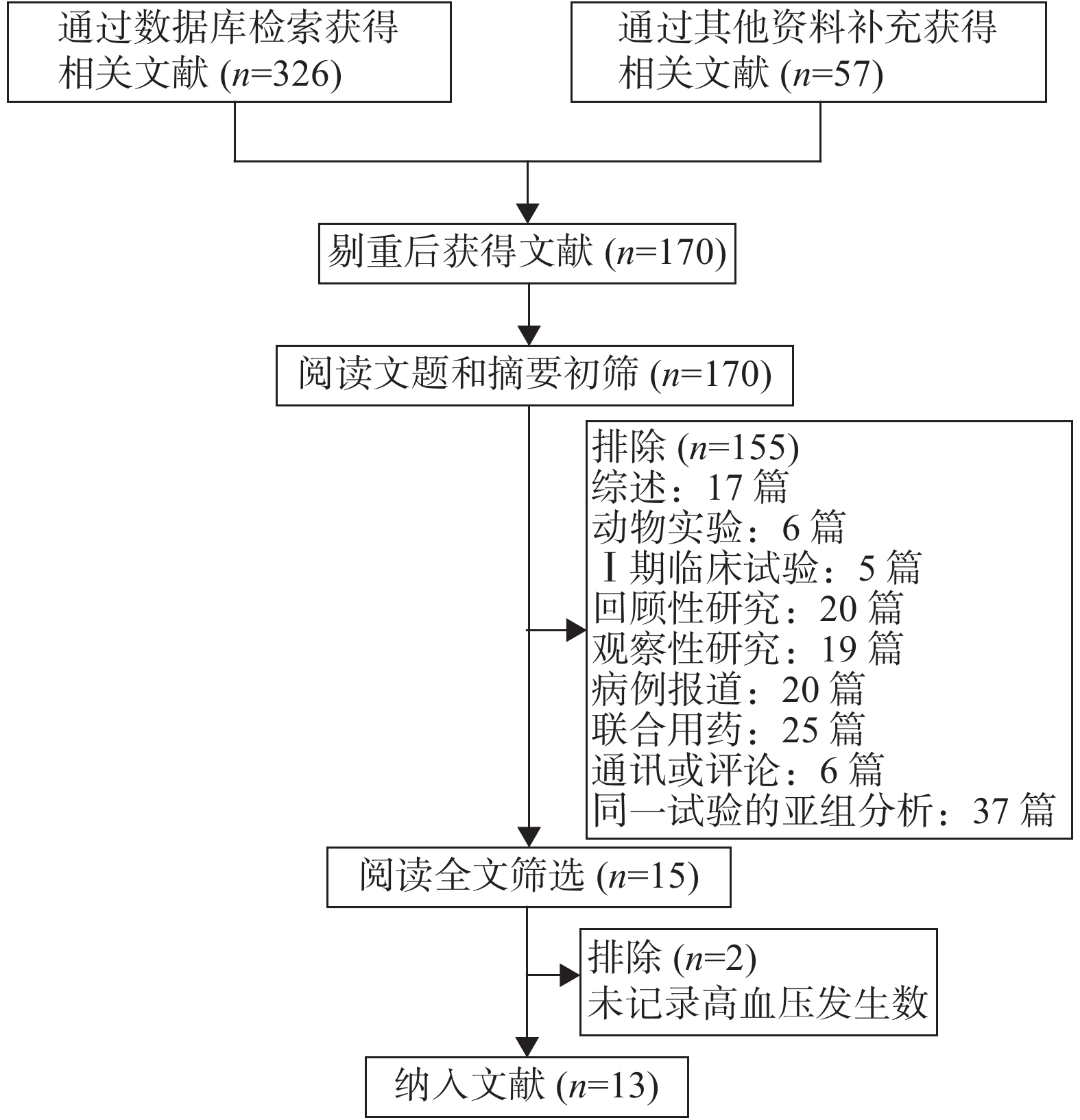
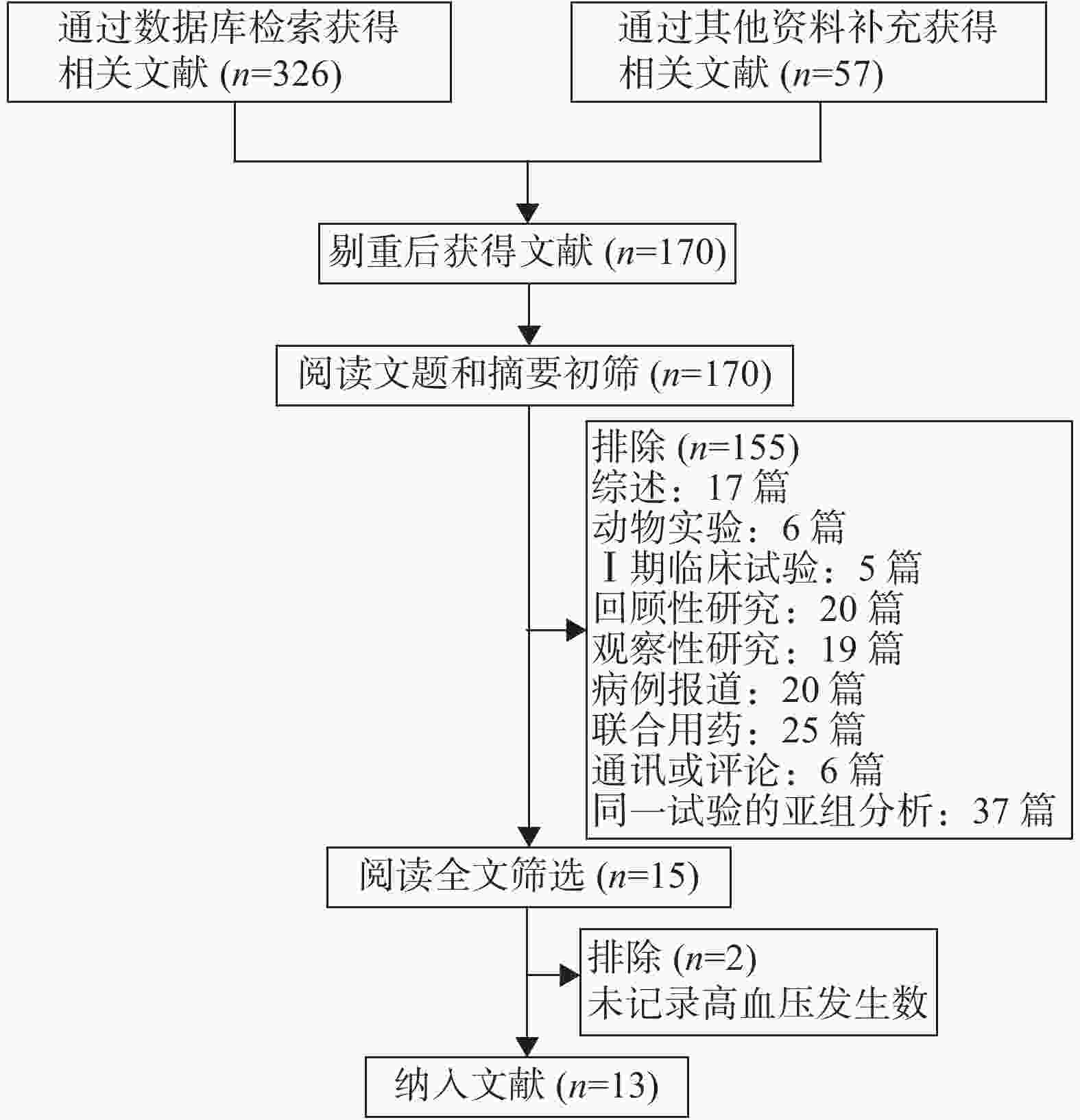
初筛后共获得文献170篇,经阅读文题和摘要后筛选出15篇文献,仔细阅读全文后,纳入符合入选标准的文献13篇。文献筛选流程见图1。
-
纳入的13项研究均为英文文献,包括6篇前瞻性单臂Ⅱ期临床试验,6篇Ⅱ期RCT临床试验,1篇Ⅲ期RCT临床试验,共纳入1387名(安罗替尼:1187人,安慰剂:200人)癌症患者进行Meta分析。纳入研究的基本特征详见表1。
纳入文献 肿瘤类型 试验设计 药物名称 患者数 高血压发生数 试验组 对照组 试验组 对照组 试验组 ≥G3① 对照组 ≥G3② Wu[8] 小细胞肺癌 Ⅱ期 单臂 安罗替尼 NA 45 NA 6 3 NA NA Han[9] 非小细胞肺癌 Ⅲ期RCT 安罗替尼 安慰剂 294 143 199 40 24 0 Ma[10] 转移性肾细胞癌 Ⅱ期 单臂 安罗替尼 NA 42 NA 19 2 NA NA Chi[11] 软组织肉瘤 Ⅱ期 单臂 安罗替尼 NA 166 NA 70 8 NA NA Sun[12] 甲状腺髓样癌 Ⅱ期 单臂 安罗替尼 NA 58 NA 23 3 NA NA Tang[13] 骨癌 Ⅱ期 单臂 安罗替尼 NA 42 NA NR 8 NA NA Shan[14] 卵巢癌 Ⅱ期 单臂 安罗替尼 NA 14 NA 8 0 NA NA Zhou[15] 转移性肾细胞癌 Ⅱ期 RCT 安罗替尼 舒尼替尼 90 43 45 12 29 11 Han[16] 非小细胞肺癌 Ⅱ期RCT 安罗替尼 安慰剂 60 57 33 6 3 0 Chi[17] 软组织肉瘤 Ⅱ期 RCT 安罗替尼 安慰剂 158 75 99 30 NR NR Huang[18] 食管鳞状细胞癌 Ⅱ期 RCT 安罗替尼 安慰剂 110 55 NR 17 NR NR Wang[19] 小细胞肺癌 Ⅱ期 RCT 安罗替尼 安慰剂 46 22 18 7 NR NR Chi[20] 甲状腺髓样癌 Ⅱ期 RCT 安罗替尼 安慰剂 62 NR 29 NR NR NR 注:NA:不适用;NR:未报道;①试验组高血压≥G3的发生数;②对照组高血压≥G3的发生数。 -
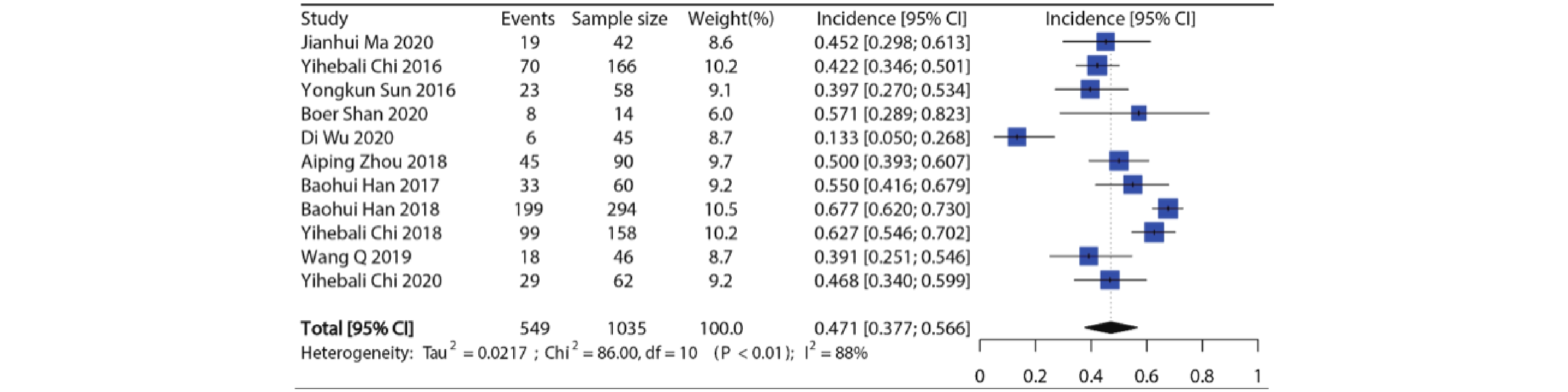
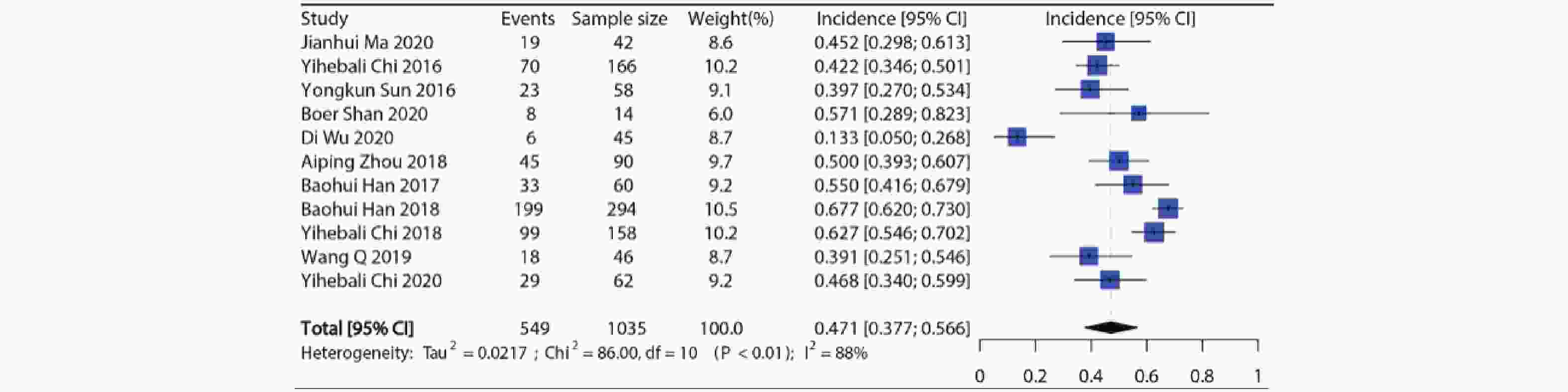
纳入11项研究共1035例病例用于分析,各个研究所有等级高血压发生率在13.3%~67.7%,发生率最低的是用于SCLC的Ⅱ期RCT临床试验[8],发生率最高的是用于NSCLC的Ⅲ期RCT临床试验[9]。Meta分析显示纳入研究之间具有异质性(I2=88%, P<0.01),因此使用随机效应模型合并,接受安罗替尼治疗的患者所有等级高血压的总发生率约为47.1%(95%CI:37.7%~56.6%),结果见图2。
-
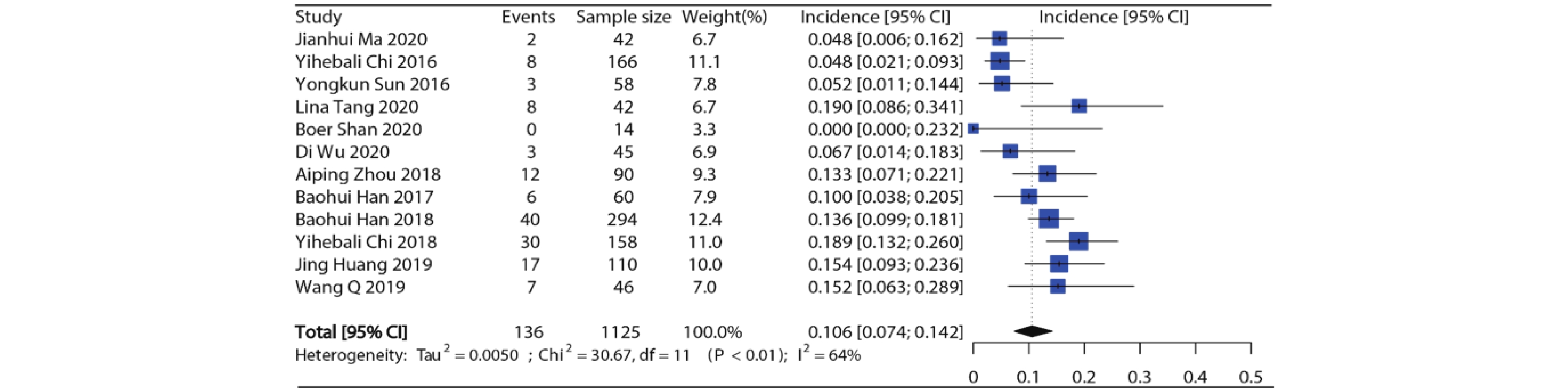
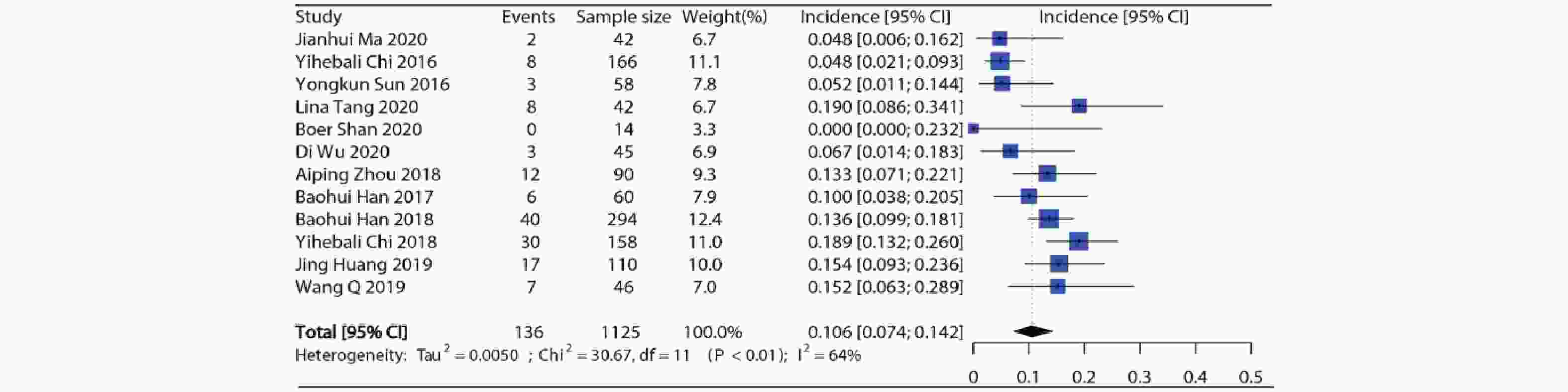
12项研究报道了接受安罗替尼的患者(合计1 125例)高等级高血压发生率,各项研究的高等级高血压发生率在0%~19.0%之间。Meta分析结果显示纳入研究之间存在异质性(I2=64%, P<0.01),因此采用随机效应模型,合并得到安罗替尼高等级高血压总发生率约为10.6%(95%CI:7.4%~14.2%),结果见图3。
-
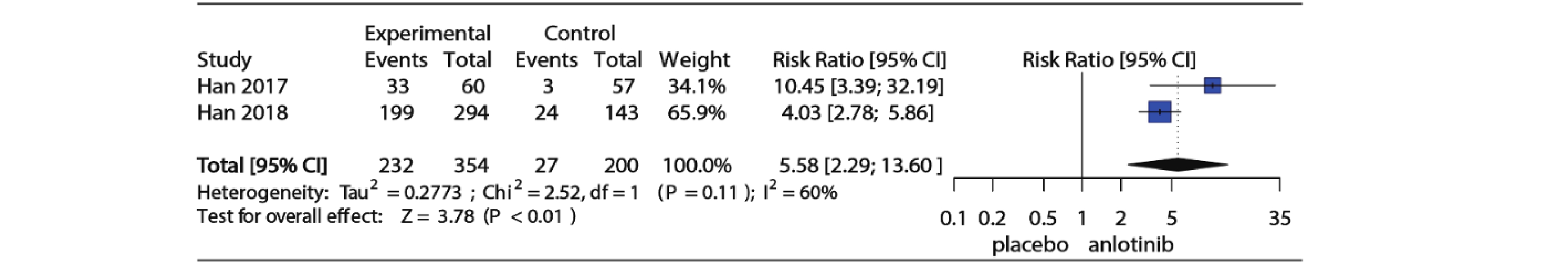
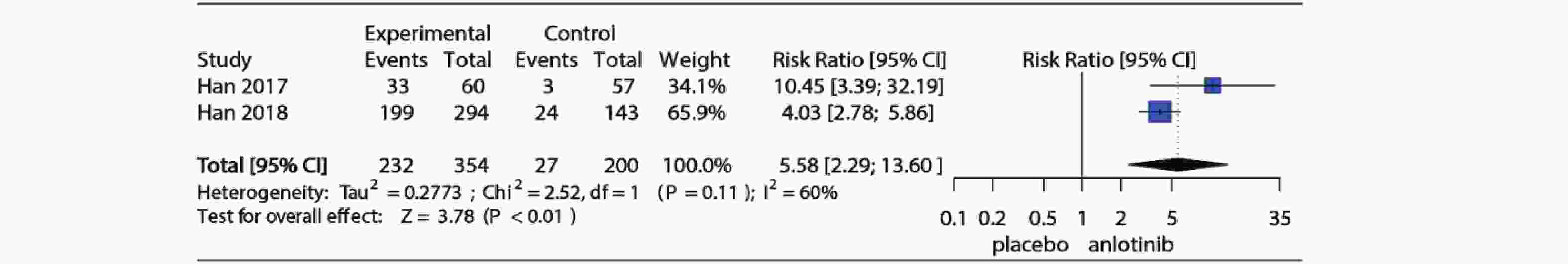
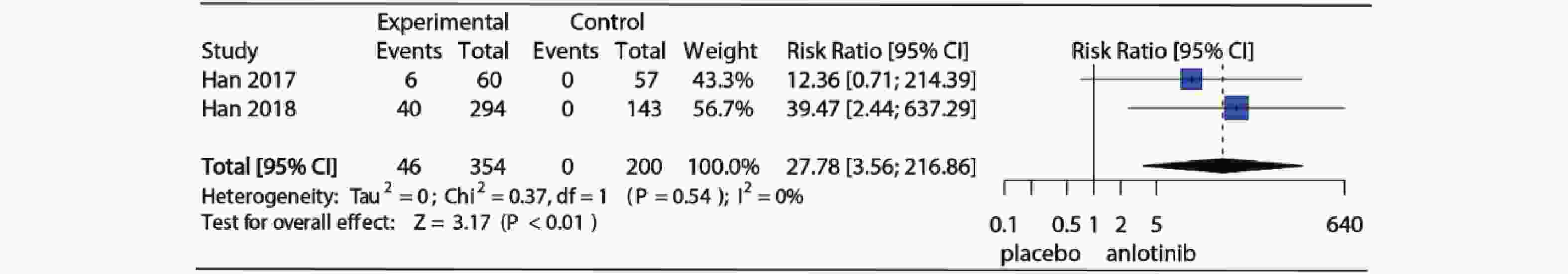
3项RCT研究详细记录了试验组和对照组高血压发生情况,2项对照组为安慰剂,1项对照组为舒尼替尼,为了明确安罗替尼与安慰剂相比高血压发生风险,仅纳入对照组为安慰剂的2项RCT研究。Meta分析结果如图4所示,I2=60%,P=0.11提示研究存在中等异质性,使用随机效应模型合并(RR=5.58, 95%CI:2.29~13.60, P<0.01)。图5为安罗替尼与安慰剂相比发生高等级高血压的相对危险度及Meta分析森林图,I2=0%,P=0.54,提示纳入的2项研究之间无异质性,采用固定效应模型合并(RR=27.78, 95%CI: 3.56~216.86, P<0.01)。
-
比较已发表的6个VEGFR抑制剂药物高血压发生率的Meta分析文献,进一步探讨了安罗替尼与其他VEGFR抑制剂高血压发生率的差异,表明安罗替尼高等级高血压发生率与阿昔替尼(RR=0.79, 95%CI:0.61, 1.02, P=0.066)和卡赞替尼(RR=0.87, 95%CI:0.67, 1.13, P=0.290)相似(见表2)。
序号 比较组别 所有等级高血压 高等级高血压 发生率(样本量) RR(90% CI) P 发生率(样本量) RR(90% CI) P 1 安罗替尼 47.1%(1035) 10.6% (1175) 2 索拉非尼 23.4% (3363) 2.91(2.51~3.37) <0.001 5.7% (3567) 1.96(1.50~2.31) <0.001 3 舒尼替尼 21.6% (4609) 3.23(2.80~3.72) <0.001 6.8% (4407) 1.62(1.30~2.03) <0.001 4 帕唑帕尼 35.9% (1242) 1.59(1.34~1.88) <0.001 6.5% (1286) 1.71(1.28~2.30) <0.001 5 凡德他尼 24.2% (1815) 2.78(2.37~3.28) <0.001 6.4% (1190) 1.73(1.28~2.34) <0.001 6 阿昔替尼 40.1% (1148) 1.33(1.12~1.58) <0.001 13.1% (1148) 0.79(0.61~1.02) 0.066 7 卡赞替尼 27.8% (1083) 2.31(1.93~2.76) <0.001 12.0% (1083) 0.87 (0.67~1.13) 0.290 注:表中RR值和P值均为安罗替尼与其他VEGFR抑制剂相比较;其他VEGFR抑制剂高血压发生率数据从已发表文献中提取。 -
Egger检验和Begg检验结果显示,纳入分析的发生所有等级高血压事件(Egger检验:P=0.050,Begg检验:P=0.243)和高等级高血压事件(Egger检验:P=0.366,Begg检验:P=0.243)的相关文献未见明显发表偏倚。
2.1. 文献检索结果
2.2. 纳入研究的基本特征
2.3. 所有等级高血压发生率
2.4. 高等级高血压发生率
2.5. 发生高血压相对风险
2.6. 不同VEGFR抑制剂之间高血压发生率差异
2.7. 发表偏倚分析
-
血管内皮生长因子(VEGF)是参与肿瘤血管生成的关键因子,目前VEGFR抑制剂是多种癌症的一线治疗药物。然而,在使用其治疗癌症的同时可能会诱发高血压,高血压不仅会增加心血管系统不良事件的发生风险,还可能累及其他系统,如导致肾脏损害、高血压性视网膜病变等[21]。
本研究共纳入1387例病例,研究结果提示接受安罗替尼治疗的癌症患者高血压发生率较高(所有等级47.1%, 95%CI:37.7%~56.6%;高等级10.6%, 95%CI:7.4%~14.2%);与安慰剂相比,安罗替尼高血压发生风险显著增加(所有等级RR=5.58, 95%CI:2.29~13.60, P<0.01;高等级RR=27.78, 95%CI:3.56~216.86, P<0.01)。因此,在临床应用过程中应重视监测患者的血压,并根据血压的分级及时进行处理。
尽管有实验和临床证据表明VEGFR抑制剂与其发生高血压有直接的关系,但具体病理生理学和分子生物学机制尚不明确。其可能的机制包括:①VEGF通过促进内皮细胞释放一氧化氮(NO)从而产生血管扩张的效应,VEGFR抑制剂则会导致NO合成减少促进血管收缩,导致外周阻力增加和血压升高[22];②内皮素-1分泌增加,导致全身血管阻力增加和高血压效应[23];③VEGF可维持毛细血管网的完整性,当受到抑制时,可能会导致毛细血管的密度下降或结构疏松从而导致高血压[24]。
目前尚无关于使用VEGFR抑制剂治疗的患者血压管理的正式指南,美国国家癌症研究所药物研究委员会的心血管毒性小组建议[25]:①在开始VEGFR抑制剂治疗之前进行正式的心血管风险评估;②在整个治疗过程中监测血压和心脏毒性,在治疗初期更应密切监测;③积极管理早期出现的高血压和心脏毒性以预防长期心血管系统损害。
VEGFR抑制剂诱导血压升高治疗药物的选择应注意避免使用非二氢吡啶钙离子拮抗剂,因为其抑制CYP450 3A4,可能导致VEGFR抑制剂血药浓度升高,加剧VEGFR抑制剂诱导的高血压[26]。应避免同时使用CYP3A4抑制剂(如维拉帕米或地尔硫䓬等)。肾损害也是VEGFR抑制剂常见的不良反应,因此,控制安罗替尼所致的高血压最常用的是血管紧张素转换酶抑制剂(ACEI)或血管紧张素II受体拮抗剂(ARB),若控制不佳,可以加用其他降压药(如钙离子拮抗剂、利尿剂、β阻滞剂等)[27]。
不同VEGFR抑制剂高血压发生率有较大的区别,因此,笔者进一步分析了使用安罗替尼与其他VEGFR抑制剂发生高血压的差异。从表2可以看出,使用安罗替尼的患者高血压发生率均高于其他大部分VEGFR抑制剂(包括索拉非尼[2]、舒尼替尼[3]、帕唑帕尼[4]、凡德他尼[5]、阿昔替尼[6]和卡赞替尼[7])。对于高等级高血压发生率,安罗替尼与阿昔替尼和卡赞替尼相似,高于其他VEGFR抑制剂。不同VEGFR抑制剂高血压发生率具有较大差异的原因可能是由于部分VEGFR抑制剂还作用于其他靶点,如安罗替尼作用于VEGFR、FGFR、PDGFR和c-kit[1],而卡赞替尼作用于VEGFR和间质表皮转化因子受体[7]。然而,缺乏安罗替尼与其他VEGFR抑制剂大样本量的头对头临床研究,而且患者的基线特征可能会影响结局指标,所以应该谨慎解释这些结果。
本研究的局限性:①本研究为Meta分析,不是基于患者的原始数据进行分析,因此可能存在一些混杂因素;②纳入研究未描述基线的高血压发病率,这可能是导致Meta分析后高血压总体发生率较高的原因之一,然而,我们使用了RCT研究的数据进行RR值的合并,从而减少误差;③纳入的研究之间存在潜在的差异,如不同的肿瘤类型等,但纳入计算RR值的临床试验的癌种均为NSCLC;④纳入研究的数量有限;⑤大多数临床试验会排除控制不佳的高血压或重大心血管疾病的患者,因此高血压的发生率和严重程度在真实世界中可能会更高。
-
本文首次对癌症患者接受安罗替尼治疗相关的高血压总体发生率和风险进行Meta分析,使用安罗替尼会显著增加发生高血压的风险,建议临床用药前进行系统的心血管风险评估,用药期间密切监测血压并根据其分级进行适当管理,特别是对于高危患者。选择治疗高血压药物时,需注意药物相互作用。安罗替尼为多靶点TKI,临床应用过程中也应关注与其他靶点相关的不良反应。仍需进一步开展大样本量的研究,以明确其可能发生的机制和与其相关的危险因素。







 DownLoad:
DownLoad: